New Pre-HSCT Treatment May Make Stem Cell Transplants a Safer Option for MS Patients
Written by |
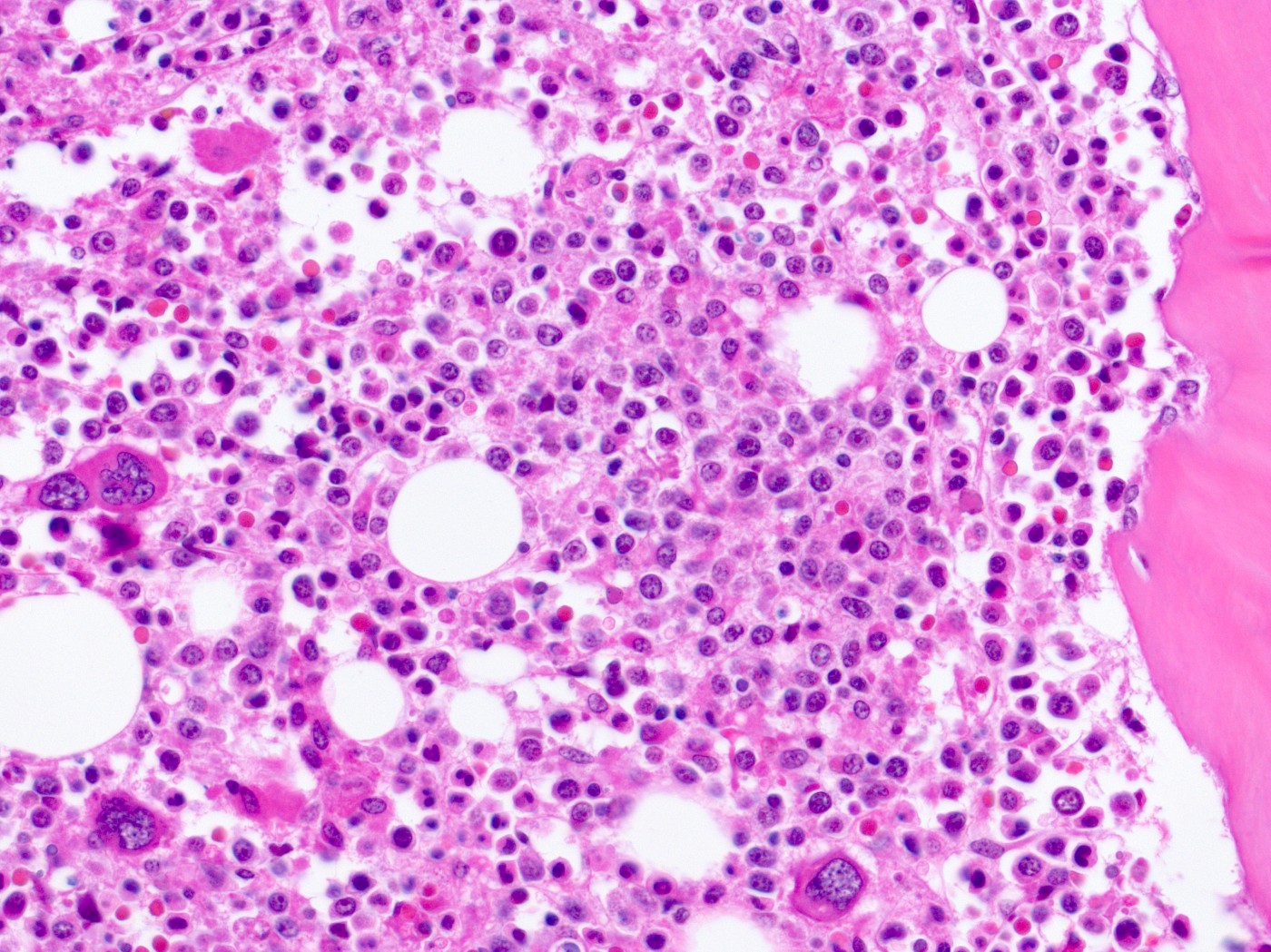
Scientists at Stanford University School of Medicine have developed a method for stem cell transplants that may do away with the need for prior systematic treatment with chemotherapy or radiotherapy. If successful, stem cell transplants could be an option for patients with multiple sclerosis (MS), an option now limited by the risk of severe toxicity triggered by the chemotherapy.
The study, “Hematopoietic stem cell transplantation in immunocompetent hosts without radiation or chemotherapy,” was the cover story of the latest issue of the journal Science Translational Medicine. The method has been successfully tested in mice, but if proven safe and effective in humans, it could truly revolutionize MS treatment, as well as the treatment of a host of other conditions.
“If it works in humans like it did in mice, we would expect that the risk of death from blood stem cell transplant would drop from 20 percent to effectively zero,” the study’s senior author, Dr. Judith Shizuru, MD, PhD, and a professor of medicine at Stanford, said in a news release.
While blood, or hematopoietic stem cell transplants, known as HSCT, have been available for MS patients for some time, health authorities in many countries — including the U.S. and EU — have not yet approved such treatments for MS. Lately, studies have supported the idea of stem cells being used for MS and a recent clinical trial found the method successful in 23 out of 24 patients.
The final patient is, however, what prevents the treatment from becoming a standard treatment for all MS patients. That person died of damage caused by chemotherapy.
Aggressive chemotherapy, needed to wipe out all the old bone marrow cells, is not easy for the body to endure. In addition to killing the old stem cells, it kills a host of other cells and can cause liver damage, destroy the ability to have children, and lead to irreparable brain damage. On a longer time scale, cancer is one of the most common side effects of chemotherapy. (Of note, the Canadian trial used a highly potent chemotherapy regimen, chosen because of its ability to cross the blood-brain barrier. But the doses were higher than those used in most HSCT clinics now making this therapy available to MS patients.)
Instead of killing all dividing cells, like chemotherapy does, Stanford researchers developed a much more precise method to get rid of old stem cells. A combination of two antibodies — one targeting a surface protein called c-Kit on blood stem cells, and another that blocks a molecule called CD47 — allowed immune cells called macrophages to destroy cells covered with anti c-Kit antibodies.
In mice, the combination rapidly cleared old stem cells, allowing for the successful transplant of new stem cells with very little side effects.
In addition to this safer method to clear the way for new stem cells, researchers also improved the transplant itself. Cells taken from the bone marrow — the ones that are used for stem cell transplants — are effectively a mix of stem cells and various immune cells from the donor. These old immune cells contribute to what is known as graft-versus-host disease, with the old cell system attacking the tissues of their new host. The researchers “purified” the donor tissue so that it contained only blood stem cells.
Most MS stem cell treatments use so-called autologous transplants, taking out the patient’s own stem cells before killing the immune system with chemotherapy. Today, successful autologous stem cell transplants may put off symptoms and disease progression for years, but many patients do relapse after transplants, and lingering immune cells in a transplant derived from the patient’s own bone marrow likely contribute to that.
“If and when this is accomplished, it will be a whole new era in disease treatment and regenerative medicine,” said Dr. Irving Weissman, MD, a study co-author and professor of pathology and developmental biology at Stanford, who is also the director of the Stanford Institute for Stem Cell Biology and Regenerative Medicine, and director of the Ludwig Center for Cancer Stem Cell Research and Medicine.


Trevor
How do I Enron for the trial in humans, I am in the UK
Sam D'Uva
How long is it forecasted to take for this procedure to be an approved method of treating MS patients?
Dana May
I would give anything to be in one of the first trials for this in US. Dr Wiessman is brilliant! I can't even describe how grateful I am for this research! How can I contact someone regarding this?
Ellie Baum
Dr. Richard Burt at Northwestern has been conducting HSCT in a clinical trial- see clinicaltrials.gov. I had HSCT 2 months ago and am already so much better it's unbelievable.
Dana
Thank you Ellie. I am not qualified for Northwestern though as I have had lemtrada. That's why I was so excited to see this that doesn't require additional chemo before receiving the stem cells. However, I am so glad that you had HACT and are already much better! Best wishes!
rob r.
lemtrada is enough. it acts virtually the same as hsct.
Dana
Really? My MS is still progressing after 2 years of treatment. Lemtrada is not 100% successful in restarting the immune system of everyone who takes it. Lemtrada does not kill all the malfunctioning lymphocytes, just a lot of them. It's unfortunate that you tell me I've had "enough" treatment without knowing my medical history or the correct success rate of lemtrada. I am hanging on by a thread for hope in future treatments.
Lenore kimmons
I am thinking about having this treatment done. Can you tell me about your experience and any issues since. Where did you go to have it done.
Courtney Wright
How do I get information on how to enter the trial for this when it starts? Or any trial for that matter.
Ellie Baum
www.clinicaltrials.gov
Craig Tyler
It is unfortunate that the HSCT treatment referenced by this article is an older form of HSCT. Most HSCT centers throughout the world have changed to a less aggressive form of chemo - virtually eliminating the mortality rate (and other side effects) without lowering the success rate of the treatment. HSCT has consistently demonstrated in many clinical trials to be the best treatment for MS.
Brian Melton
You are correct. HSCT is the ONLY hope currently. When this theory moves into clinical trials most of us will be beyond repair. I hope most of our MS warriors understand this.
Ellie Baum
Yes I'm not understanding this 20% mortality rate....are they talking about the mice? Because I'm only aware of the one person expired in the small Ottawa trial with more advanced MS cases due to complications but not anyone in any other trial.
Brian Melton
Yes they are out of there minds. There have been a few deaths along time ago but that was using poor prognosis patients and total body eradication.
Michele Morales
Craig Taylor you are correct. The mortality rate for nonmyeloblative HSCT is less than 1℅. I received HSCT in Puebla, Mexico with Dr. Ruiz a world renowned hemotologist Mayo Clinic alumni.
Mary E. Miller
Hi Michael,
Can you give me a website, etc. for Dr. Ruiz? It is for my sister. She applied for the clinical trial at Northwestern but was considered too advanced.
Thank you,
Mary
Lenore kimmons
What kind of results did you have.
rob r.
this stanford news is fantastic!! It will improve hsct which is already the best thign out there for ms!!
Shazia Aslam
Which country is the safest to have this treatment .i have ms and want this treatment
Wandert van Zwol
Hi,
My name is Wandert and live in the Netherlands.
I would be more than willing to try this in a trail!!!
If possible please send info!!!
Cara
Too bad we'll never see it happen. I'm sick of mice stuff. When I see that, it normally means it'll be looked at and maybe approved within the next 20 years.
Michael Guzzetti
You are so right Cara,I myself am tired of hearing about mice,I had stem cell done in 2014 and it was a very expencive wast of money.And yes it is true that most of us won't be around when they do find something to help us.In my opinion there will still be hope for all of us.As of now I have had SPMS since 2012 DX 1999 it's been a long hard road,but I try to make the best of it,and I urge you
and every one suffering to do the same,never ever give up.
Ken Bloom
Where did you have your HSCT in 2014?
Thanks.
Many of us are searching for HSCT options and there appears to be a lot of places worldwide that will gladly take your money with no data to support their treatment or positive results that can be measured. Lots of claims and patient comments but no data.
Shazia Aslam
I'd like to take part in a trial please let me know how I can do that
Meg
http://www.nbc29.com/story/32854359/virginia-mom-heading-to-mexico-to-beat-ms
Read it
Philip Longford
Do NOT get conned! So many 'clinics' springing up, across the world, offering 'Stem Cell Treatment'. Unless they are, (at the moment), also doing the chemo (HSCT), everyone seems to say, it is useless.
I will be the first to sign up, IF I am confident there is, at least, some chance of success.
Hi Philip, my personal view is to only go with HSCT with chemo. It's chemo that is crucial, not stem cells.
Sherry
I believe this works. I was accepted in Mexico but sadly had to turn it down do to money and caretaker issues ????
Please feel free to join my page on Facebook MS stories
Love n love that to you all ????????
Hi Sherry, so sorry that you had to turn down HSCT in Mexico. Like you, I believe it works. This particular story, however, is about the development of a drug to replace the chemotherapy element of HSCT. Availability of such a drug, if it can be proved to work, is some considerable way off.