Study Examines Gadolinium Deposits in MS Patients’ Brains, But Still Can’t Determine Relationship with Disease Severity
Written by |
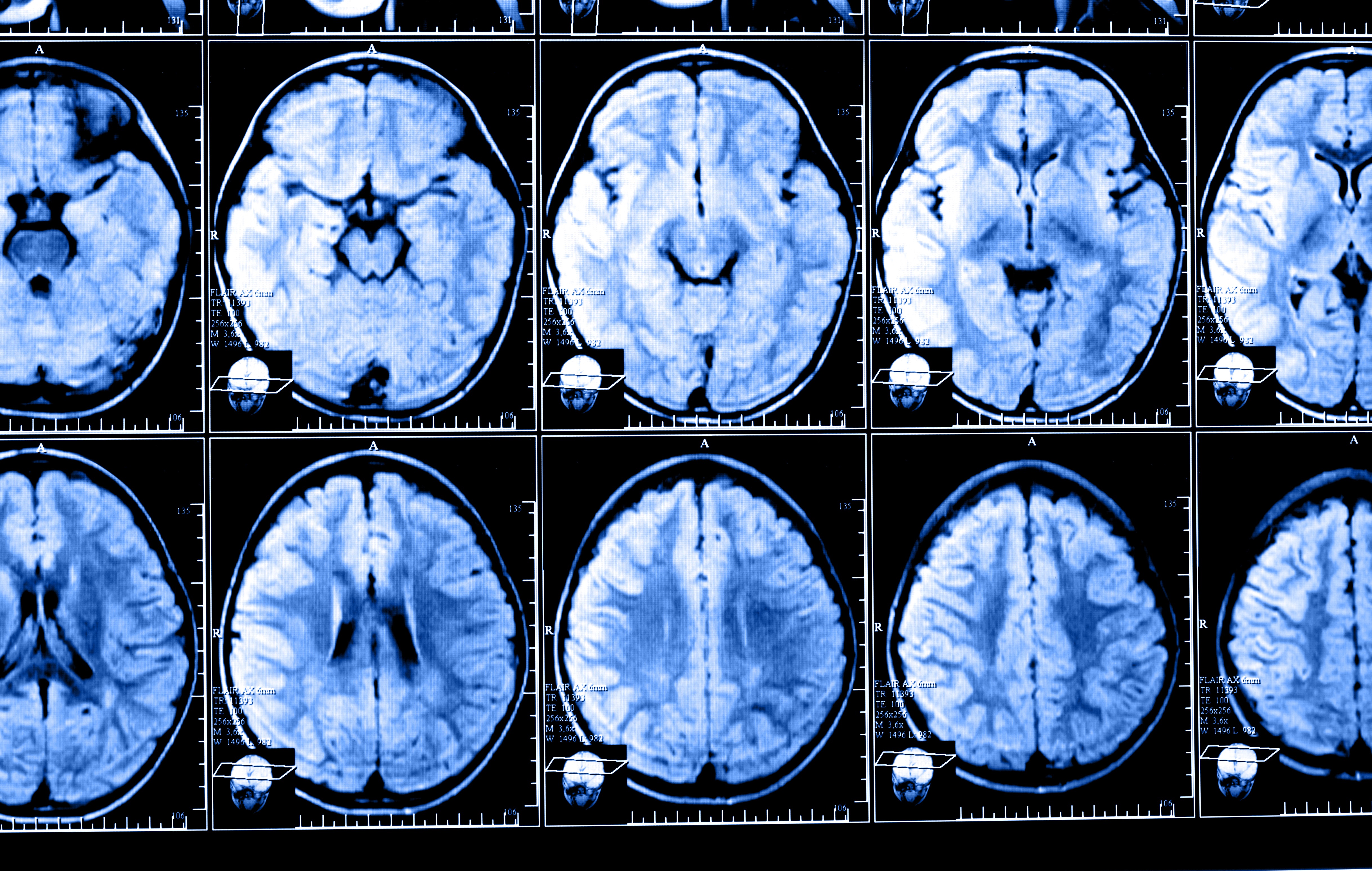
The use of gadodiamide, a gadolinium-based contrast agent (GBCA) often used to help clinicians visualize brain structures in magnetic resonance imaging (MRI) scans, leads to the accumulation of gadolinium in certain regions of multiple sclerosis (MS) patients’ brains early in the course of the disease, a study has found.
However, the relationship with disease severity remains unclear.
Those findings, in the study “Cumulative gadodiamide administration leads to brain gadolinium deposition in early MS,” were published recently in Neurology, the medical journal of the American Academy of Neurology (AAN).
In its natural state, gadolinium is highly toxic. For that reason, in order to be used in clinical practice as a contrast agent, gadolinium needs to be joined with other molecules, giving rise to different types of GBCAs.
There has been controversy in the MS community about the use of GBCAs as contrast agents. That is primarily because some of these agents are associated with gadolinium accumulation in the brain of MS patients who frequently undergo MRI brain scans to monitor the progression of their disease, raising the concern of toxicity and disease worsening.
Due to these concerns, four GBCAs already have been banned in Europe, and in 2017 the U.S. Food and Drug Administration (FDA) has issued a recommendation stating the use of GBCAs should be kept to a minimum.
Several studies have shown that patients who frequently undergo brain scans, and are injected with contrast agents, develop gadolinium depositions in the brain. However, none of those studies monitored long-term brain alterations associated with the use of GBCAs in MS patients.
In the recent study, a group of researchers from the Jacobs School of Medicine and Biomedical Sciences at the University at Buffalo, New York, examined the progression of brain alterations occurring in MS patients since experiencing their first symptoms, up to five years after being diagnosed.
The large-scale longitudinal study involved 203 MS patients, who were followed at the Buffalo Neuroimaging Analysis Center (BNAC) of the University at Buffalo from 2003 to 2016. All patients had received identical doses of gadodiamide during each MRI session performed at the Buffalo General Medical Center. The study also included 262 healthy individuals used as controls.
At a mean follow-up period of 55.4 months (4.5 years), study participants received an average of 9.2 injections of gadodiamide.
Results showed that at follow-up, almost half of the MS patients (49.3%) had areas of high-gadolinium intensity in the dentate nucleus (a brain region responsible for controlling voluntary movements and cognition), while none of the controls had the same type of high-intensity gadolinium depositions in the same region.
In other brain regions, such as the globus pallidus (a region that controls voluntary movements), MS patients also had areas of gadolinium deposition of higher intensity compared to controls.
However, investigators failed to find a clear association between the presence of high-intensity gadolinium depositions and MS severity.
Interpreting the findings
Based on the results, the team concluded that brain “gadolinium deposition in early MS is associated with lifetime cumulative gadodiamide administration, without clinical or radiologic correlates of more aggressive disease,” they wrote.
“This study is one of the first to investigate the longitudinal association between well-established clinical and MRI outcomes of disease severity and gadolinium deposition,” said Robert Zivadinov, MD, PhD, said in a press release written by Ellen Goldbaum. Zivadinov is a professor in the department of neurology, director of the BNAC, and lead author of the study.
“The study didn’t find any correlation between deposition in the brain and clinical or MRI outcomes, such as accumulation of lesions, brain atrophy or disease severity, at least in the first five years of the disease,” Zivadinov added. “Over the 4.5 years of follow-up, we didn’t find that GBCA deposition contributed to patients being more disabled.”
Nevertheless, researchers did find that MS patients who had received more than eight injections of gadodiamide tended to have a higher number of brain lesions, as well as more severe brain atrophy (shrinkage), compared to those who had less than eight gadodiamide administrations.
“Therefore, we cannot completely rule out that gadolinium deposition may have an impact on disease progression or clinical outcome,” Zivadinov said, adding that the findings from the study “should be incorporated into a risk-versus-benefit analysis when determining the need for GBCA administration in individual MS patients.”
Interestingly, researchers also found that male patients were more prone to have gadolinium depositions than female patients. This unusual finding, according to Zivadinov, might be because men typically receive higher doses of contrast agents, as the dose is based on the individuals’ body weight, which may increase their chances of developing gadolinium depositions.


Jose Luis Flor Acosta
In 2015 I entered the Medical Imaging Center in Hoffman Estates Illinois 2500 W. Higgins Rd. They used GAD on me and since have lost weight, 100 lbs, convulse when I sleep or lay down and my eyesight has worsened, not sure if its GAD or something else.
haslie kemp
I have had 11 since 2005 and even with all the symptoms of MS, diagnosed 2010 with no lesions but by spinal tap, there is a question if I have MS or not. Some of the symptoms of Gad toxicity are similar to MS.
john carney
I think you have a much bigger problem than you think . The only problem is it will take more than 50 years and millions of destroyed lives before the geniuses in medicine figure all this out . GE shredded all the evidence before the MRI was released. Countries around the world have already discontinued most contrast dyes .....how many diseases do you need to create .I could go on about antibiotics and vaccines ....are they using this poison gadolinium here too ?
Loralyn Conover
If gadolinium is banned in Europe then why are we still using it in America? Why does America always have to be the last one to do the right thing?
Betty
Did the gadolinium deposit in the healthy patient's brain? and what percentage if any, developed any type of disorder or GDD that didn't previously have any problems or MS. I'm speaking of movement disorders, trouble swallowing, cognative issues, pain, swelling, etc.
I ask because I am one of those people who was affected.
Sherry L. Wood
I got Sepsis and worst headache and neckache shortly within days after contrast in MRI on I have MS.
Jonathan kelly
NO.... PUT IT IN YOUR BODY AND SEE!!!? If u have others health problems gadolinium tends to inhance those problems
Rodney N Smith
I had an cat-scan did in April a full body scan. They shot the dye in my IV. My body was cold and it started to burn as it with through my body,made me feel like I had to go pee. My head started to have severe head pain. When the scan was over,my head was having a massive headache. I was dizzy. I thought that was ok . But it wasn't. My blood pressure at the time low after that scanning it was high. I do take blood pressure pill. It did kept my pressure low. Now my bones hurt in my legs and fingers, my hands and ankles are swollen. I need to know what's going on with me.
Kym
I could not pull the full study up online , were all patients gadolinium levels tested b 4 & after having additional MRIs? Knowing Bayer company is on the advisory board & funds research for Buffalo medical school & they manufacture controversial contrast agents, like magavist, r the results favorable for the company because of their funding? When medical schools r taking money from top pharmaceutical companies & depending on that money for research do their results favor these companies?
kym zanmiller
I am allergic to this contrast, my first MRI it was used and I was itchy and flushed, the 2nd one I was also itchy, flushed and it was difficult to breathe and I was scared because you have to lie still in the MRI tube. My neurologist told me that gladium allergies are cumulitive and if it was used as a contrast again it make kill me!
They should make another contrast so that I would get more complete reports