Australian Secondary Progressive MS Clinical Trial Tests New Microparticle Therapy
Written by |

 New Zealand and Australia-based Innate Immunotherapeutics Limited, a biotechnology company with offices in Sydney and Auckland, has designed and manufactured a unique immunomodulator microparticle pharmaceutical technology. This technology can be used to induce the human immune system designed to fight certain cancers and infections, or modulate certain immune mechanisms implicated in autoimmune diseases such as Multiple Sclerosis (MS). They say the same technology can be used in designing better vaccines to potentially treat or prevent diseases such as influenza, cancer, malaria, or tuberculosis.
New Zealand and Australia-based Innate Immunotherapeutics Limited, a biotechnology company with offices in Sydney and Auckland, has designed and manufactured a unique immunomodulator microparticle pharmaceutical technology. This technology can be used to induce the human immune system designed to fight certain cancers and infections, or modulate certain immune mechanisms implicated in autoimmune diseases such as Multiple Sclerosis (MS). They say the same technology can be used in designing better vaccines to potentially treat or prevent diseases such as influenza, cancer, malaria, or tuberculosis.
There are two main forms of MS: an early “relapsing-remitting” stage of disease and a later, more disabling “secondary-progressive” stage of disease (SPMS). Worldwide, 30 percent of all MS sufferers have SPMS, and there are currently no approved disease modifying drugs for the safe and effective ongoing treatment of this highly disabling form of the disease, which causes walking, hand, eyesight, and cognitive function disabilities.
Innate Immunotherapeutics is currently recruiting subjects in Australia who have Secondary Progressive Multiple Sclerosis (SPMS) for its phase 2B trial of its experimental therapy MIS416, a biologically derived novel immune modulator that can target both the regulatory functions and the defensive (pathogenic) functions of the innate immune system. MIS416 targets myeloid cells, a sub-set of innate immune cells not currently targeted by existing or other ‘in-trial’ MS drugs.
Myeloid cells have only recently been recognized as a significant potential therapeutic target in SPMS. Myeloid cells have the capacity to remodel the deregulated immune activity, which is an important part of the disease process in SPMS. These same cells, remodeled in the correct fashion, can also promote neuro repair pathways critical to slowing or reversing disability in SPMS.
The study, which is being administered by the Western Australian Neuroscience Research Institute (WANRI), was recently approved by the Bellberry Human Research Ethics Committees (HREC) — a national, private not-for-profit organization providing streamlined scientific and ethical review of human research projects across Australia — allowing the clinical trials to commence. HREC’s mission is to promote and improve the welfare of research participants and the quality, efficiency and effectiveness of the research itself.
In August, WANRI received approval to commence patient recruitment for Innate Immunotherapeutics’ Phase 2B trial of MIS416 in patients with SPMS. WANRI has been selected by Innate as the study’s lead site, due to its strong focus on investigating the causes and improving the therapy and management of patients suffering from multiple sclerosis. The Institute has a strong focus on providing the best clinical management for multiple sclerosis available.
“We are really pleased to be leading this trial. SPMS affects 30% of the MS population at any moment in time, and there are no approved long term effective treatment options for these patients. We all hope that MIS416 might be the drug to address this urgent need,” says Dr. Allan Kermode, clinical Professor of Neuroimmunology at WANRI.
[adrotate group=”4″]
The Phase 2B trial will recruit up to 90 SPMS patients with the secondary progressive form of multiple sclerosis across eight investigational trial sites in Australia. The trial is a double blinded randomized study, where 60 patients will receive MIS416 and 30 will receive a placebo. Patients will be treated once weekly for 12 months. The primary goal of the trial is to determine the efficacy and safety of MIS416 compared to patients treated with the placebo. As part of the study, patients will also report their own health status quarterly, as previous study results have shown significant and sustained reductions in pain and fatigue.
Innate says that the previous non-placebo controlled MIS416 studies found that 80% of patients with SPMS had shown a 30% or greater improvement in at least one measure of their MS-related symptoms. Notably, patient stakeholder groups such as Multiple Sclerosis Research Australia (MSRA) and the United States National Multiple Sclerosis Society (US MS Society) have both expressed strong support of Innate’s pursuit of an effective treatment for SPMS. The experimental medication has been studied and used as part of the ongoing compassionate use program in Australia, and revealed promising results in studies led by researchers at Victoria University of Wellington in which researchers demonstrated that MIS416, developed originally to treat the relapsing-remitting form of MS, is efficient in the treatment of secondary progressive multiple sclerosis as well. However, the research team has not fully understood what makes the therapy effective.
A Phase 1B human study in patients with primary progressive or secondary progressive MS (SPMS) was completed in October 2011, and a follow-up ‘dose confirmation’ Phase 2A study in patients with secondary progressive MS was completed in July 2012. Results from these studies met or exceeded expectations, leading to the launch of the current study.
How MIS416 Works in Treating SPMS
The microparticle at the core of Innate Immunotherapeutics’ technology provides a unique delivery system for a suite of both known and novel immune system triggers or modulators. By attaching selected modulators to the microparticle, these triggers can be delivered reliably to specific cells of the immune system.
Target diseases or applications for drugs developed using this approach include:
• Auto-immune diseases such as Multiple Sclerosis;
• Certain cancers that are known to be immune-sensitive, such as prostate, colon, & renal cancer; and
• Preventive vaccines for malaria and tuberculosis, and therapeutics vaccines for cancers.
The lead drug candidate developed using this technology, MIS416, is a construct of the microparticle and naturally occurring, non-immunogenic, cytosolically-active TLR-9 and NOD-2, and has demonstrated safety and efficacy in a number of different disease or injury models. When administered, these microparticle/ligand complexes are readily absorbed by the cells of the reticulo-endothelial system, with cellular activation occurring only after microparticle internalization and subsequent biodegradation of the microparticle and release of the ligands. Signal transduction pathways activated by these ligands act in concert to activate a broad range of innate immune cell subsets that are central to the development of innate and adaptive anti-tumour responses.
In vitro studies have shown that MIS416 has specific immunomodulatory effects on dendritic cell, monocyte / macrophage, Natural Killer (NK) and NKT-cell cytokine secretion patterns, in parallel with enhancement of a broad range of soluble and cellular tumouricidal mechanisms such as granule, Fas, TRAIL and TNF-a.Due to the significant degree of unmet medical need, Secondary Progressive Multiple Sclerosis has been selected as the initial clinical target for MIS416.
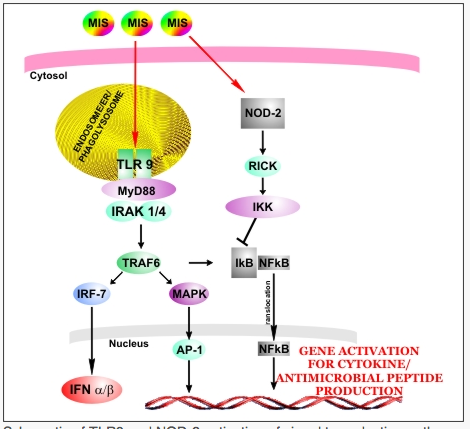
Schematic of TLR9 and NOD-2 activation of signal transduction pathways Activation of both of these pathways correlates with the early production of NFkB-dependant inflammatory cytokines and chemokines that act locally, serving to recruit cells and act as immune maturation stimuli. These mechanisms underpin the co-ordinated activation of a range of innate immune responses — image credit Innate Immunotherapeutics
Unlike most pharmaceutical agents (drugs or monoclonal antibodies), immunomodulators like MIS416 do not act directly on the target (cancer tumor, infectious agent, damaged nerve), but instead switch on powerful disease fighting mechanisms that form part of the human immune system. The immune system is a collection of biological barriers and processes that protects against disease by identifying and killing external threats such as infectious agents (bacteria, viruses, parasites, other pathogens) and internal threats such as cancer tumor cells. To function properly, the immune system needs to distinguish between threats and the body’s own healthy cells. When this ability to distinguish between non-self and self breaks down, the immune system can attack healthy cells resulting in one of several autoimmune diseases.
When administered as a distinct agent, Innate Immunotherapeutics says MIS416 is a potent activator of broad but well characterized innate immune responses. The immune system comprises several layers of defense. The first line of defense consists of physical barriers, such as the skin and mucous membranes that line the digestive, respiratory, and reproductive tracts. For infection to occur, pathogens must first breach this physical barrier. When such a breach does occur, the innate immune system is the next line of defense — ‘innate’ because all animals naturally possess it from birth. The surveillance cells of the innate immune system firstly recognize the signature (or pattern) of an invading pathogen and then activate appropriate attack cells or mechanisms to clean out the invader. This same surveillance and response process also works for cells that go bad, e.g. early stage cancer cells. If the innate system is overwhelmed, the adaptive immune system is triggered, providing the last, but often the most potent layer of immune defense. The cells that form part of the adaptive response (antibodies and killer T-cells) must be custom-made to match the pathogen and so the process is relatively slow, but once designed, these cells can be made in huge quantities to overwhelm the threat. Once the system is adapted to recognize and destroy a particular invader, it remembers that invader, and can then react more quickly next time the invader is encountered.
The innate system is pivotal to the successful operation of the entire human immune system. It recognizes trouble, provides an immediate general (or non-specific) defense and if required, gives permission to the adaptive system to respond. Importantly, it also regulates the overall reaction to ensure that the level of response is appropriate to the degree of the threat. By activating and/or regulating important innate system mechanisms, Innate Immunotherapeutics’ microparticle immunomodulator technology presents a new and effective way to safely fight infections, certain cancers, and to treat select autoimmune diseases.
 Innate Immunotherapeutics’ Chief Scientific Officer Gill Webster obtained postdoctoral research experience investigating the development of immune cell-based cancers and mechanisms underlying cancer gene regulation pathways was obtained at the University of Glasgow, UK and the University of Dundee Wellcome Trust Biomedical Research Centre, UK. As an expert in the field of Flow Cytometry, she held senior scientific research positions at Cyclacel, Dundee, U.K. and Genesis Research and Development, New Zealand for several years prior to joining Innate Immunotherapeutics.
Innate Immunotherapeutics’ Chief Scientific Officer Gill Webster obtained postdoctoral research experience investigating the development of immune cell-based cancers and mechanisms underlying cancer gene regulation pathways was obtained at the University of Glasgow, UK and the University of Dundee Wellcome Trust Biomedical Research Centre, UK. As an expert in the field of Flow Cytometry, she held senior scientific research positions at Cyclacel, Dundee, U.K. and Genesis Research and Development, New Zealand for several years prior to joining Innate Immunotherapeutics.
Dr. Webster says that there is a need to be able to adequately modulate the host immune system following stem cell grafting because they become susceptible to rejection when differentiating into somatic lineages. There is also a need to temper the inflammatory response associated with tissue engineering and other repair techniques that can lead to scar tissue formation. Dr. Webster notes that MIS416 can control overt inflammation as well as antigen-specific T cell activity associated with graft rejection thereby assisting the repair process.
Preliminary findings also indicate that MIS416 therapy may mobilize the endogenous stem cell axis in whole organisms, which may also contribute towards successful engraftment. Dr. Webster says MIS416 demonstrated an excellent safety profile in the successfully completed human phase 2a clinical trial in secondary progressive Multiple Sclerosis patients in New Zealand. According to regular safety blood tests performed, the patients are able to receive weekly or fortnightly injections of MIS416 on a long-term and continuous basis, and there was no significant dose intolerance or cumulative toxicity reported. Animal toxicology studies have demonstrated that acute, twice weekly (for 4 weeks), and once weekly (for 26 weeks) i.v. administration of MIS416 is well tolerated and at doses up to 20 times the target human dose, observed effects were not explicitly adverse and were the result of the drug’s extended pharmacological effects.
New Zealand medicines regulator, Medsafe, approved the manufacture of MIS416 as a medicine in early 2008 — approval that means the drug is being made in accordance with the internationally agreed rules of Good Manufacturing Practice (“GMP”). As part of the GMP licensing process, formal stability studies have been performed on samples of MIS416 subjected to multiple freeze-thaw cycles, cold storage (36 months at 2-8 degrees C), and room temperature storage. These studies have confirmed that the bioactivity of MIS416 is stable across the whole range of storage temperature tested for periods in excess of 24 months.
Innate Immunotherapeutics says proof-of-principle studies have demonstrated the immunomodulator micro particle technology to have efficacy in diseases or applications including: autoimmune conditions — specifically Multiple Sclerosis and cancer — examples include metastatic breast, lung, and melanoma disease; infectious disease — examples include influenza and plague and as an adjuvant in an experimental anthrax vaccine; and, radiation injury induced neutropenia. In addition, the microparticle as a vaccine carrier and adjuvant has been evaluated in both malaria and tuberculosis vaccine development programs and in the development of cancer treatment vaccines.
Patient stakeholder groups such as Multiple Sclerosis Research Australia (MSRA) and the United States National Multiple Sclerosis Society (US MS Society) have both expressed strong support of Innate’s pursuit of an effective treatment for SPMS. MSRA CEO Dr Matthew Miles says: “Australia has a strong cohort of world-leading MS neurologists who are highly experienced clinical trial investigators and strongly committed to supporting the development of new treatments for their patients.”
Sources:
Innate Immunotherapeutics
Western Australian Neuroscience Research Institute (WANRI)
Bellberry Human Research Ethics Committees (HREC)
Consortium for Medical Device Technologies
Multiple Sclerosis Research Australia (MSRA)
Image Credits:
Consortium for Medical Device Technologies
Multiple Sclerosis Research Australia (MSRA)



Kyla Mathers
I have already sent my information asking for more information and received an email stating someone would be in touch but still have heard nothing. Could I please ask for more information AGAIN?
Maggie
First of all: Worldwide, 30 percent of all MS sufferers have Secondary Progressive MS. I do not believe that percentage is at all accurate and here's why. I was 23 when I was dx'd with RRMS in 2002. All of my inflammatory, aka, central nervous system attacks happened before 2005. My doctors would still to this day have me labeled as Relapsing Remitting if I hadn't insisted they relabel my type of MS and level of disease. It's because they cannot prescribe most of the expensive big pharma drugs to people with Progressive MS and most likely insurance won't pay for them either. Right now I have an EDSS of 6.5 and I haven't had any immune system attacks in years. I have old damage, old lesions and my immune system works great and has for years. It seems like MS could be a result of an unknown event whether it be vascular, trauma, an infection, toxins, etc. that set off an immune response to attack. Once that defensive immune response has been alleviated your left with degenerative nerve damage. That's the area of progressive MS that needs to be addressed.
Hitesh B Parmar
Hi
I am Hitesh Parmar from state Gujarat, India
My father is suffering from SPMS and PPMS has started before 10 years. He is 100% disable now especially with both the legs and left hand and even he is not able to make any movement of the body himself. I would like to participate him on the research test for the same.
We are assuming cause of PPMS is he had internal injury in the brain in road accident in year of 1988 and PPMS started in 2005 due to cloating in the blood.
Please contact me for further process of testing for the same.
Regards,
Hitesh