CorTechs Labs And Novartis Partner On Solutions for Quantifying Brain Volume In MS Patients
Written by |

San Diego based medical software developer CorTechs Labs announced that it has entered into a partnership agreement with multinational pharmaceutical maker Novartis Pharma AG. The two companies will collaborate in further development of Cortechs’ powerful NeuroQuant breakthrough 510(k) cleared software brain volume quantification device that targets identification, measurement and tracking of brain volume loss in multiple sclerosis (MS) patients. NeuroQuant is designed to make quantitative MRI measurement and analysis of MRI images of the human brain and fully automated MRI post-processing capabilities to the physician’s desktop or mobile device, a routine part of clinical practice.
A NeuroQuant report provides neurologists, radiologists, and clinical researchers with a convenient and cost-effective means to quantify atrophy of brain structures to help diagnose a variety of brain disorders, including conditions such as Alzheimer’s disease, epilepsy, multiple sclerosis and traumatic brain injury. It enables clinicians to conduct sophisticated, automated, accurate, and consistent measurements of the whole brain, the left and right thalami and the lateral ventricles using data from 3D T1 MRI images, and targets identification and tracking of neurodegeneration in these sub-cortical structures.
Non-invasive, inexpensive and accurate, NeuroQuant is used to verify Alzheimer’s in patients 50-95, assessment of epilepsy and multiple sclerosis in patients 18 and older, traumatic brain injury in NFL players, boxers and soldiers, and more. Cortechs’ technologies provide physicians with the objective information they need to confirm or refute a projected diagnosis.
NeuroQuant is available both online and as an installed software solution, provides neurologists and radiologists with objective data on brain volume loss and the brain’s changes over time compared with normative values.
 “We’re pleased to partner with Novartis to advance the clinical care and assessment of multiple sclerosis worldwide,” says CorTechs Labs’ CEO Guri Stark CorTechs Labs’ CEO Guri Stark, “We believe this partnership will empower clinicians with objective quantitative data, while continuing NeuroQuant’s growth momentum. This, in-turn furthers our vision to establish NeuroQuant as the standard measurement solution for all brain disorders, including Alzheimer’s disease, epilepsy, multiple sclerosis and traumatic brain injury.”
“We’re pleased to partner with Novartis to advance the clinical care and assessment of multiple sclerosis worldwide,” says CorTechs Labs’ CEO Guri Stark CorTechs Labs’ CEO Guri Stark, “We believe this partnership will empower clinicians with objective quantitative data, while continuing NeuroQuant’s growth momentum. This, in-turn furthers our vision to establish NeuroQuant as the standard measurement solution for all brain disorders, including Alzheimer’s disease, epilepsy, multiple sclerosis and traumatic brain injury.”
NeuroQuant is the first FDA-cleared and CE marked (a mandatory conformity marking for certain products sold within the European Economic Area) medical device capable of automatically detecting and quantifying human brain atrophy. It provides a convenient and cost-effective means by which neurologists, radiologists, clinical researchers and imaging centers can quantify brain structure atrophy.
NeuroQuant automatically segments and measures volumes of the hippocampus, ventricles and other brain structures and compares these volumes to normal values. Reducing subjectivity in the diagnosis process, Cortechs’ software brings quantified measurements of neurodegeneration directly to the specialist’s desktop.
For data input, NeuroQuant takes a high-resolution 3D T1-weighted sagittal, non-contrast-enhanced MRI scan from a disk or auto-routed from a scanner or PACS server, and after adjusting for distortions, the software identifies and segments multiple brain structures. It then measures their volumes and compares the volumes to a normative database adjusted for age, sex and cranial volume.
That process takes only a few minutes, with no user intervention, and yields a new, full-volume, spatial corrected and automatically labeled dataset, along with printable patient reports. These reports contain absolute and relative volumes of the hippocampus, temporal horn and other structures in a DICOM-compatible format.
A NeuroQuant locally installed system on a computer integrates seamlessly with existing PACS solutions. CorTechs can install the solution on a Mac provided by the customer. The Installed System of NeuroQuant is currently compatible with Mac OS X (10.6 and above) workstations. NeuroQuant® works on all Intel-based Macs, optimally with at least 2 GB of RAM. Additional RAM and CPU speed will decrease processing time.
NeuroQuant Online allows users to access the solution immediately with no installation required, and at a very low risk. The web-based version is already used by some of Cortechs’ largest customers and partners and enables secure on-site or off-site web access. Users can log onto NeuroQuant Online anytime, on any Internet-connected device, from virtually anywhere. Individual scans are submitted for analysis on an as-needed basis, with online technical support always available. Individual MRI series can also be uploaded from any computer or device connected to the Internet.
Along with being able to track brain volumes over time, CorTechs has begun to develop tools intended to transform assessment of and diagnose Alzheimer’s disease, epilepsy, and traumatic brain injury.
“Over 150 institutions around the world are currently benefitting from the use of NeuroQuant,” the company says in a statement. “It is allowing medical professionals to have an exciting new, state-of-the-art tool that directly quantifies atrophy of the brain. The response has been nothing short of remarkable. It is the only FDA cleared medical device to provide fully-automated quantification of regional brain volumes at a fiscally conscience valuation. This allows a comparison to a normative database — adjusted for specific criteria (age, gender, cranial volume) — to support a physician’s impression of the presence of neurodegenerative illness. This invaluable software coupled with the ability to track volumes across time has begun to transform the assessment of Alzheimer’s disease, epilepsy, traumatic brain injury and other neurodegenerative disorders. It also allows an enhanced ability to determine the efficacy of therapies and treatments.”
[adrotate group=”4″]
Examples of the sort of information NeuroQuant can provide include:
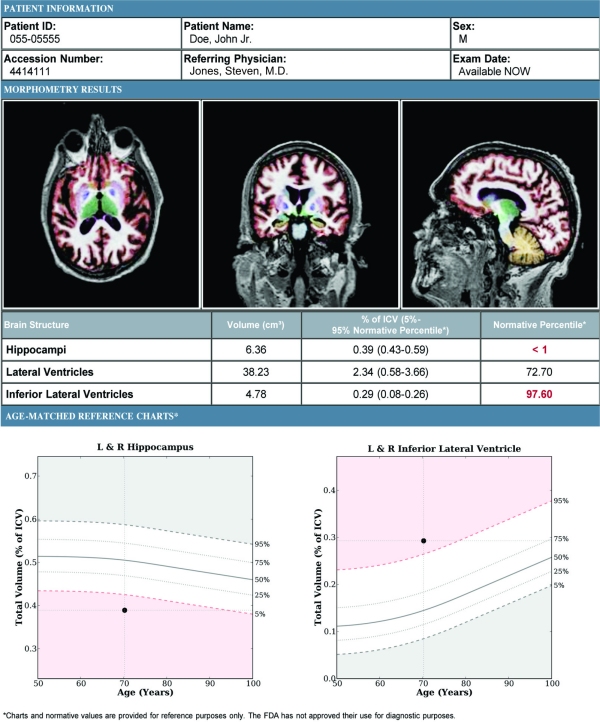 Age-Related Atrophy Report
Age-Related Atrophy Report
– A report containing the absolute and relative volumes of the hippocampus and inferior lateral ventricular structures that typically show significant atrophy in dementia patients. Resulting values are automatically compared to age-appropriate reference distributions. The interpreting physician can select representative slices and add a comment in the provided text box to be included in the report.
Hippocampal Volume Asymmetry Report AsymmetryReport-normal-hipp-abnormal-asym.pdf
– The NeuroQuant Hippocampal Volume Asymmetry Report presents the absolute and relative volumes of the left and right hippocampus in tabular form and on age and gender matched reference charts. The interpreting physician can select representative slices and add a comment to be included in the report.
General Morphometry Report
– A report providing the absolute and relative volumes for the right and left side of the following brain structures: Forebrain Parenchyma, Cortical Gray Matter, Lateral Ventricle, Inferior Lateral Ventricle, Hippocampus, Amygdala, Caudate, Putamen, Pallidum, Thalamus and the Cerebellum. It also provides an Asymmetry Index (defined as the difference between left and right volumes divided by their mean).
Color-Coded Brain Segmentation
– A 3D MR series enabling closer inspection on a PACS or other DICOM viewer.
Longitudinal Study Report
– A longitudinal assessment of the same patient at multiple scan intervals to examine regional atrophy over time.
For more information, visit:
https://www.cortechslabs.com
or
https://www.wholebrainatrophy.com
Sources:
CorTechs Labs
Novartis Pharma AG


Leave a comment
Fill in the required fields to post. Your email address will not be published.