UB Team Finds a Way to Make Stem Cells into Myelinating Cells
Written by |
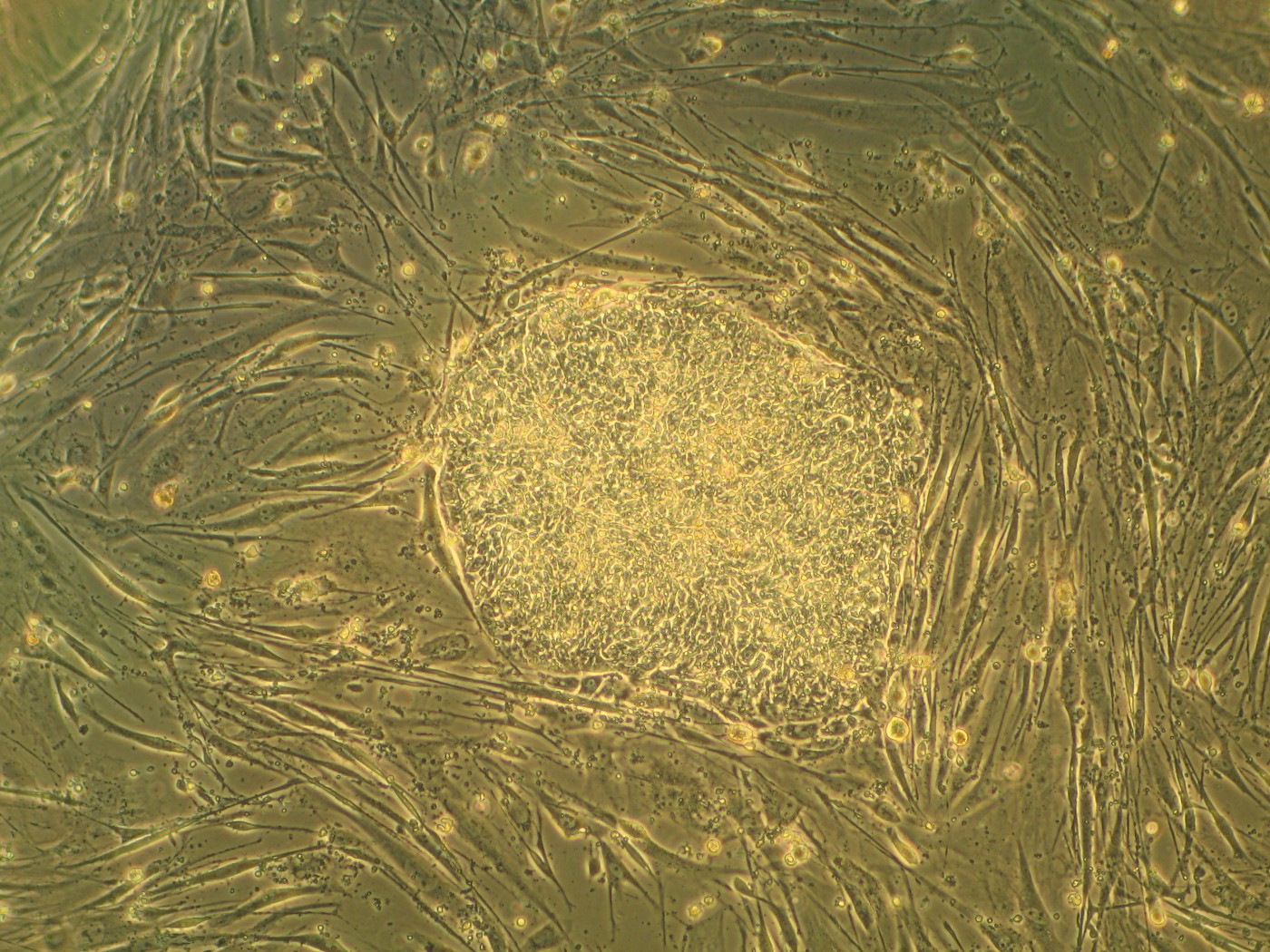
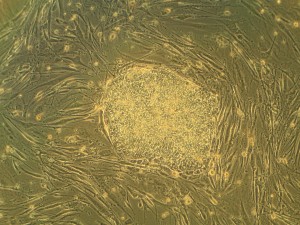 Stem therapy to treat multiple sclerosis may benefit greatly from a new study published in Proceedings of the National Academy of Sciences by the laboratory of Fraser Sim, Ph.D., assistant professor of Pharmacology and Toxicology at University of Buffalo’s School of Medicine and Biomedical Sciences. Dr. Fraser’s team discovered that transcription factor (proteins that induce gene expression) SOX10 is a master switch for inducing myelination in oligodendrocytes of the brain.
Stem therapy to treat multiple sclerosis may benefit greatly from a new study published in Proceedings of the National Academy of Sciences by the laboratory of Fraser Sim, Ph.D., assistant professor of Pharmacology and Toxicology at University of Buffalo’s School of Medicine and Biomedical Sciences. Dr. Fraser’s team discovered that transcription factor (proteins that induce gene expression) SOX10 is a master switch for inducing myelination in oligodendrocytes of the brain.
“Now that we have identified SOX10 as an initiator of myelination, we can work on developing a viral or pharmaceutical approach to inducing it in MS patients,” said Dr. Sim in a news release. “If we could create a small molecule drug that would switch on SOX10, that would be therapeutically important.”
Dr. Sim’s team, including lead author Jing Wang, a doctoral candidate at University of Buffalo, studied fetal brain stem cells for transcription factor expression. They identified transcription factors turned on in oligodendrocyte progenitors, but not neural progenitors. “We narrowed it down to a short list of ten transcription factors that were made exclusively by oligodendrocyte progenitor cells,” said Dr. Sim. “Among all ten transcription factors that we studied, only SOX10 was able to make the switch from neural progenitor to oligodendrocyte progenitor cell.”
Remarkably, SOX10 was capable of differentiating oligodendrocytes both on the lab bench and in a model of human leukodystrophy. These results indicate increasing SOX10 expression in stem cells will enhance the efficacy of human transplant therapy.
Stem cell therapies are being investigated by a number of companies, such as StemGenex and ImStem, to treat multiple sclerosis. A key obstacle to stem cell treatments is inducing cells to differentiate appropriately and in a proper time frame. According to Dr. Sim, it can take at least a year to differentiate enough stem cells into oligodendrocytes to treat one multiple sclerosis patient. “The ideal cell to transplant is the oligodendrocyte progenitor cell,” said Dr. Sim.
The process of differentiation is slowed by the number of steps it takes. Considering an autologous example, a patient’s skin or blood cells must be transformed into induced pluripotent stem cells, which then must be differentiated in neural progenitors before they can be differentiated into oligodendrocytes. “Ideally, we’d like to get directly to oligodendrocyte progenitors,” said Dr. Sim. “The new results are a stepping stone to the overall goal of being able to take a patient’s skin cells or blood cells and create from them oligodendrocyte progenitors.”
[adrotate group=”4″]
Results from the study could have implications in non-stem cell transplantation-based therapies, as well. “If we could boost the regeneration step [following immune system attack] by facilitating formation of oligodendrocytes from progenitor cells, then we might be able to keep patients in the relapse remitting stage of multiple sclerosis, a far less burdensome stage of disease that the later, progressive stage.”
Dr. Sim is funded by the New York State Stem Cell Science award led by SUNY Upstate Medical Center. Research on this project will test stem cell implantation to enhance myelination in multiple sclerosis patients.

Leave a comment
Fill in the required fields to post. Your email address will not be published.