Role of SARM1 Enzyme, Key Player in Neurodegeneration, Detailed in Studies
Written by |
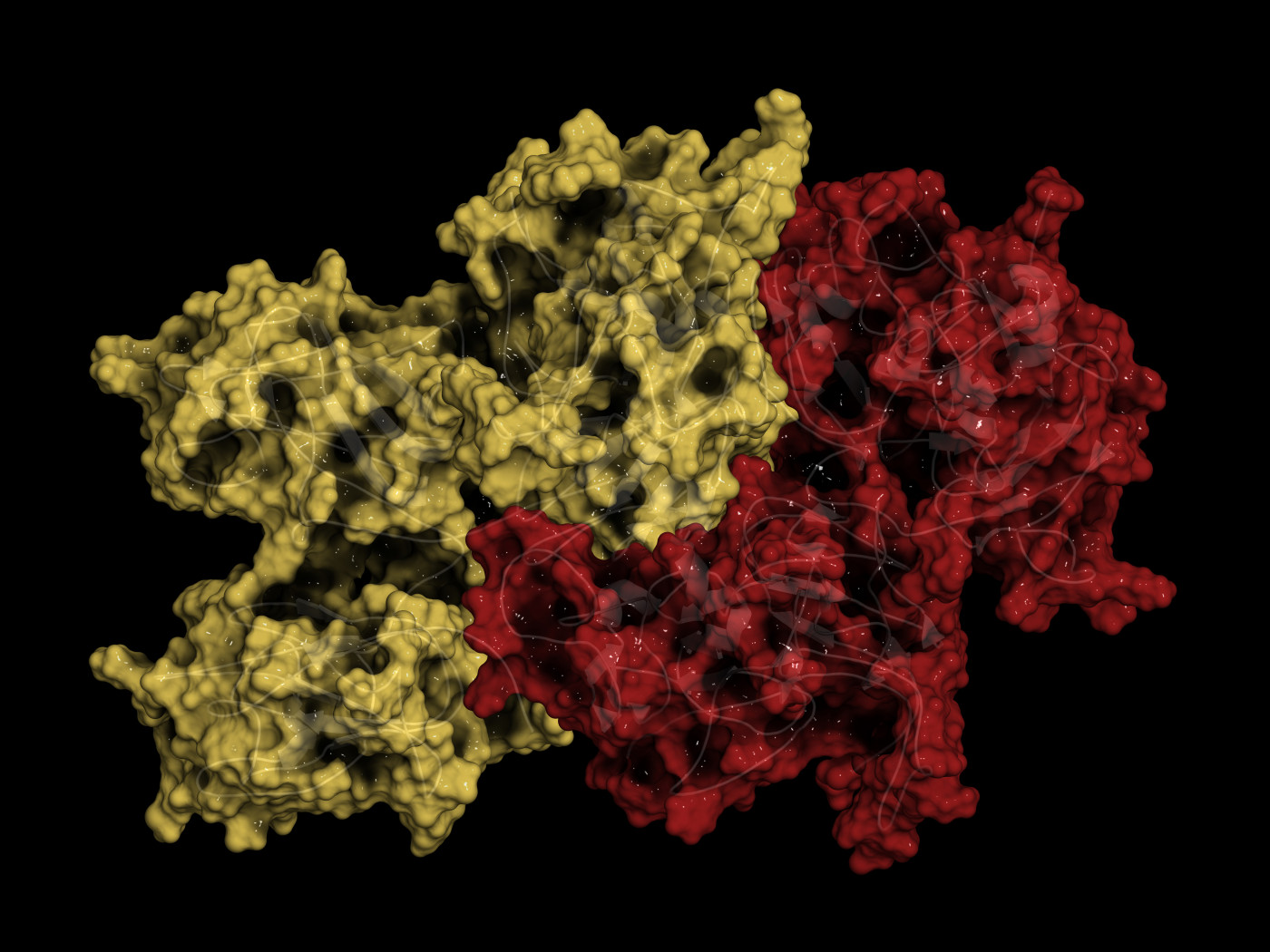
Two new studies delve into the structure of SARM1, an enzyme that plays a key role in nerve cell degeneration, and are expected to aid the development of targeted therapies for neurodegenerative disorders such as multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), and peripheral neuropathies.
SARM1 is known to be one of the drivers of damage mechanisms that induce the degeneration of axons, extensions of nerve cells (also known as neurons) that are responsible for the transmission of the electrical signals within the central nervous system (brain and spinal cord).
In a previous study published in the journal Neuron, researchers at Disarm Therapeutics reported that SARM1 was the main driver of axonal degeneration and that its harmful enzymatic activity might be targeted by a new class of small chemical compounds.
The company presented a poster showing that blocking SARM1 activity protected axons from damage, and reduced the levels of two biomarkers of SARM1 activity and neurodegeneration in neurons cultured in the lab. The poster was presented during the 2018 Neuroscience conference in San Diego.
Disarm has now announced the publication of two studies in the journal Science detailing the impact of the enzyme’s structure on its activity and function. (Enzymes are proteins able to process chemical changes in specific molecules, either transforming them into other active chemical entities or breaking them down. Often, they require a non-protein co-factor to work as intended.)
In the first study, “NAD+ cleavage activity by animal and plant TIR domains in cell death pathways,” researchers focused on exploring the structure of two domains of SARM1 — the sterile alpha motif (or SAM) and the toll/interleukin-1 receptor (or TIR) — which are very important for its enzymatic activity.
They found that both domains are necessary for the enzyme to be able to breakdown nicotinamide adenine dinucleotide in its oxidized form (NAD+), a metabolic cofactor that exists in all living cells and whose main function is to support the chemical reaction that occurs when the enzyme binds to its targets. Without NAD+, the enzyme will not be able to function.
Australian researchers working with the Disarm team found that it is this NAD+ cleavage activity that is responsible for the activation of chemical cascades that lead to axonal degeneration and neuron cell death. According to the researchers, these findings may open the door to designing and developing compounds that interfere with the normal structure of SARM1, impairing its activity, and consequently preventing axonal degeneration.
“SARM1 is a potentially transformative drug target with broad therapeutic applications for patients, and Dr. [Bostjan] Kobe’s research provides critical insights into the structure of SARM1 and the mechanism by which it mediates axonal degeneration,” Alvin Shih, MD, CEO of Disarm Therapeutics, said in a press release. “This work enables Disarm’s efforts to develop drugs for neurological diseases with substantial unmet medical need.”
In a second study, “TIR domains of plant immune receptors are NAD+-cleaving enzymes that promote cell death,” researchers found that SARM1’s TIR domain is highly conserved among plants and animals, and is one of the major mediators of innate immunity (general response against harmful micro-organisms) and cell death response in plants.
“This important recent publication … reinforces the critical NADase function of TIR domains in driving cellular degeneration across species,” Rajesh Devraj, PhD, chief scientific officer and co-founder of Disarm Therapeutics. “We look forward to continuing our partnership … as we develop SARM1-targeted treatments for severe neurological diseases.”

Leave a comment
Fill in the required fields to post. Your email address will not be published.