Structure and Workings of Receptor Tied to Inflammation and Cell Death Seen for First Time
Written by |
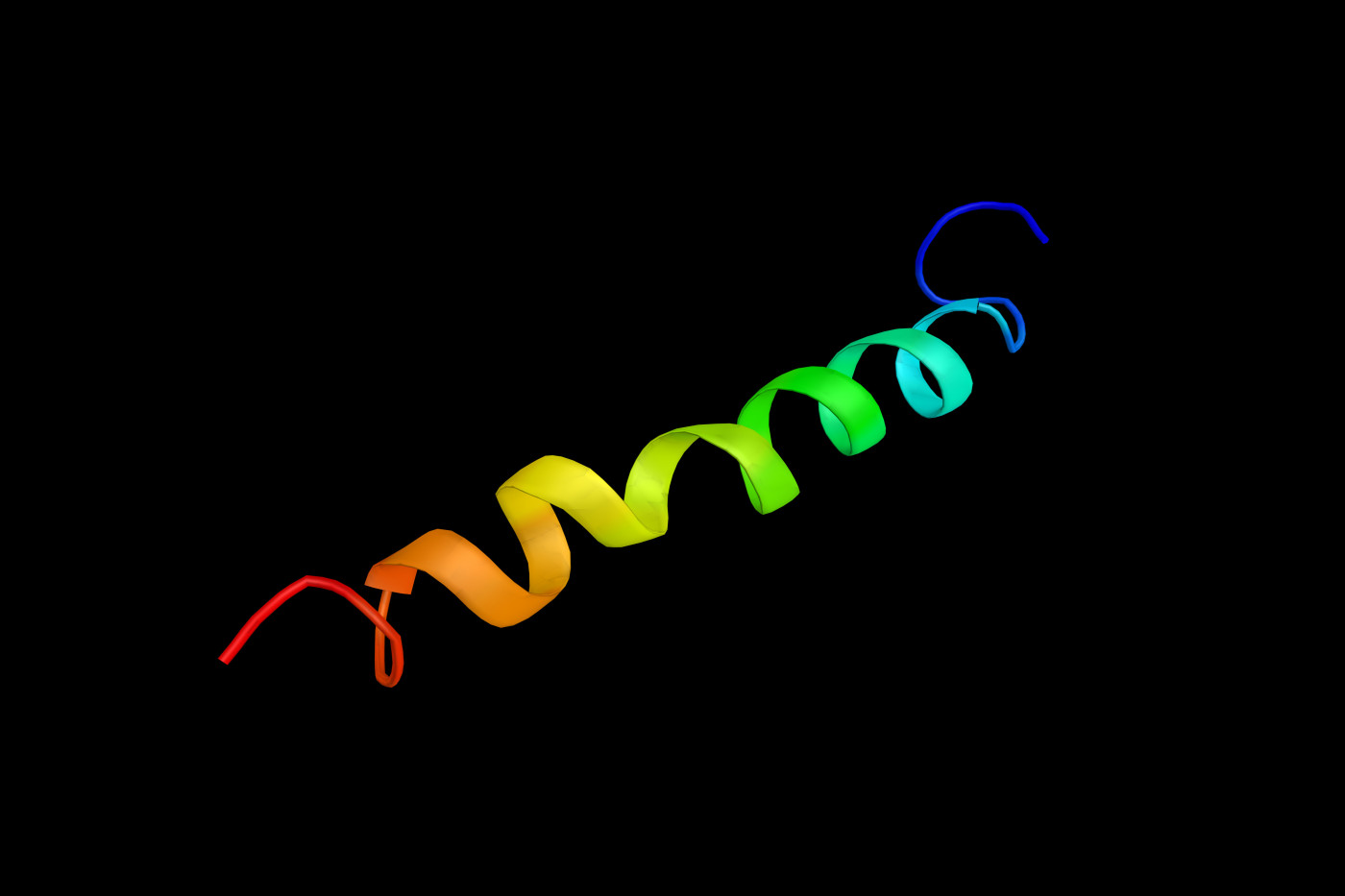
Scientists observed for the first time the molecular structure of the P2X 7 receptor, a protein that plays a key role in cell death, inflammation, and cancer progression. This breakthrough could lead to new ways of treating multiple sclerosis (MS), coronary artery disease, cancer, and inflammation.
The findings were reported in the study, “Full-Length P2X7 Structures Reveal How Palmitoylation Prevents Channel Desensitization,” published in the journal Cell.
P2X receptors are proteins found on the membranes of cells that work as channels, transporting molecules with a positive electrical charge (cations), such as calcium and sodium, in and out of cells. A portion of each of these receptors can be found outside of cells (extracellular domain), while the rest are inside (cytoplasmic domain).
The P2X 7 receptor, a member of the P2X family, has been studied as a therapeutic target because of its association with certain disease processes, such as inflammation, cancer’s spread, and neurodegeneration.
Although researchers anticipated the structure and function of the P2X 7 receptor would be very different from that of other P2X receptors, due to its unique ability to stay open indefinitely after being activated, they did not know which aspects distinguished its molecular structure from other receptors in this family.
Investigators at Oregon Health & Science University (OSHU) used a technique called cryogenic electron microscopy (cryo-EM) to see for the first time the 3D molecular structure of the cytoplasmic domain of the P2X 7 receptor in action.
In this way, they found the cytoplasmic domain of P2X 7 was bound to several groups of fatty acid molecules, a chemical modification known as palmitoylation (the addition of palmitoyl groups).
They watched as the indefinitely open channel allowed cations to freely enter cells, triggering a series of disease processes that led to cell death.
However, when they removed these fatty acid molecules from the cytoplasmic domain, they found that P2X 7 was no longer able to remain open indefinitely, stopping the charged molecules from accumulating inside cells and sending out signals that served as triggers.
“Our structures provide first insights into the architecture and function of a P2X receptor cytoplasmic domain,” the team wrote.
“Researchers have known ligand-gated ion channels are modified by palmitoyl groups, but we had never directly observed it until now,” Steven Mansoor, MD, PhD, an assistant professor of medicine (cardiovascular) at the OHSU School of Medicine and the Knight Cardiovascular Institute, and a corresponding author of the study, said in a news release written by Franny White.
“Our finding could be used as a model of how palmitoylation modifies other ion channels,” Mansoor added.
In addition to being bound to palmitoyl groups, the cytoplasmic domain of P2X 7 was also bound to guanosine, a nucleoside that is thought to have neuroprotective properties, the researchers reported.
According to the team, next steps in research will include exploring the role of these palmitoyl groups and guanosine in the context of certain diseases, including how they could be targeted as a new way of treating disorders associated with the P2X 7 receptor.

Leave a comment
Fill in the required fields to post. Your email address will not be published.