UK Team to Test Robot-assisted Exercise to Improve Balance in MS Patients
Written by |
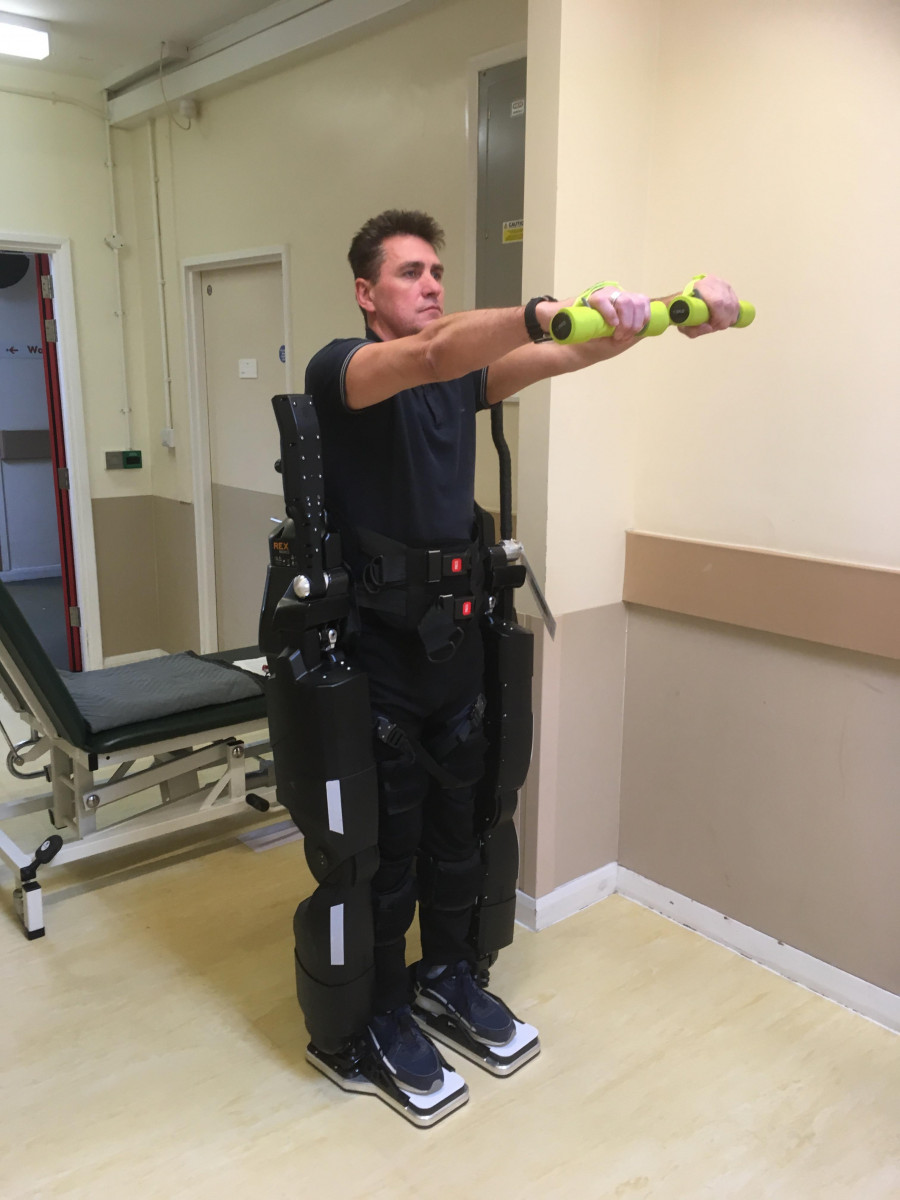
A patient completes balance and strengthening exercises using the Rex robot.
Researchers at Kent and Canterbury Hospital, U.K., are recruiting 20 patients with multiple sclerosis (MS) who can walk at least eight meters with a walking aid to test the effectiveness of a five-week exercise program using assistive robotic technology.
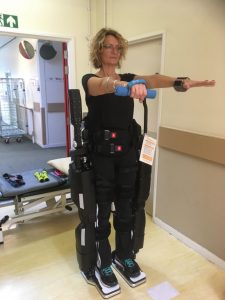
A patient completes balance and strengthening exercises using the Rex robot. (Photo courtesy of East Kent Hospitals University NHS Foundation Trust.)
The Rex device — made by Rex Bionics — is a maneuverable exoskeleton that straps to and supports a patient’s feet, legs, and core, while leaving their hands free to function normally.
If the results are positive, principal investigator Mohammed Sakel, FRCP, physician and director of Neurorehabilitation Service at East Kent Hospitals University NHS Foundation Trust, plans to seek funding to complete a large-scale, randomized, controlled clinical trial.
Sakel told Multiple Sclerosis News Today that while MS does lead to muscle weakening, impairment is exacerbated by a lack of exercise. That, he says, is due to fear of falling. “That’s where I’m using the robotic device. That will take away the fear of the fall and they can actually do the exercise.”
Patients aged 18 to 80 years first will be screened over the phone, and then in person at the hospital. Once accepted into the trial, participants will visit the hospital once a week to complete balance and core-strengthening exercises using the robotic equipment under the supervision of the neuro-physiotherapist.
The primary outcome will be measured after five weeks, using the Berg Balance Scale, a widely-used 14-piece assessment of fall risk for patients with MS, as well as other conditions that affect balance.
The study, called RAPPER IV (Robot-Assisted Physiotherapy Exercises with Rex IV) is building on results from RAPPER III, which Sakel presented at the American Congress of Rehabilitation (ACRM) annual conference in October 2018.
RAPPER III
The 10-patient RAPPER III study showed that participants, who all had moderate-to-severe MS disability as measured by the Expanded Disability Status Scale (EDSS), were able to transfer themselves into the apparatus within two minutes, though five participants required supervision or assistance.
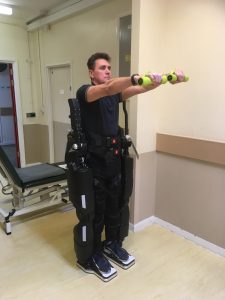
A patient completes balance and strengthening exercises using the Rex robot.
“That’s one of the markers I wanted to see, whether it is very cumbersome [to get into the device,]” said Sakel.
Patients completed four weekly appointments using the machine.
All 10 patients were able to go from sitting to standing to walking within three minutes with supervision.
Secondary outcomes showed statistically significant improvements, including: an increased range of motion of the right knee by a mean of nine degrees, and a decrease in ankle spasticity as measured by decreased inversion (mean of 8 degrees on the left ankle), decreased dorsiflexion (mean of 0.4 degrees on both left and right), and decreased plantar flexion (mean of 0.9 degrees on the left.) There was no statistically significant improvement for the left knee’s range of motion or right ankle inversion.
Forty percent of patients experienced clinically significant improvements in balance.
Participants also saw a decrease in their scores on the MS Impact Scale-29 (a measure of how much the disease affects their lives) by a mean of 26 points, and quality of life as measured by EQ-5D-5L (a survey of perceived function and mood; the higher the value, the worse the patient’s quality of life) by a mean of 0.17 points.
Competence (mean of 0.5 points), adaptability (mean of 0.7 points), and self-esteem (mean of 0.7 points), as assessed by the Psychosocial Impact Assistive Devices (PIAD) Scale, all improved in the MS patients analyzed. Of note, the PIAD scale is used to measure a person’s experience with a range of assistive technologies.
Expanding on RAPPER III
The RAPPER IV trial is expanding to include patients with mild MS disability, and also will add several new secondary outcome measures. Scientists will electronically measure how the patients distribute their weight when they walk, and determine if they are walking safely and recruiting new muscles.
A focus group will ask about the experiences of the patient as well as a spouse, close friend, or caretaker. Lastly, a six-month follow-up will test if gains are sustained over time.
The device is not be limited to use only by patients with MS. It also has been used to aid in rehabilitation in patients with spinal cord injuries and stroke.
Sakel imagines a hospital could own one or two devices that patients can visit to use for rehabilitation. Over time, he said, “[patients] won’t be needing the device so frequently.”
For more information about the study, one can contact Karen Saunders, a consultant clinical & research neuro-physiotherapist at East Kent Hospitals University NHS Foundation Trust, by emailing [email protected], or calling 07920785768.

madeline newton
this is so wonderful ...i agree with all that you are doing and it would really help us regain our muscle tone it would be so great ...keep up the work and maybe someday we all can run and ride a bike again...love it...
Ineke Driece
It all looks too good to be true! It sounds that this is what I have been waiting for. Keep up the good work.
Brad X Pan
Hope it will be available for MS patients. I am a MS patient and participated the clinical at TIRR in Houston, TX, USA which is very helpful. I am planning to buy it whenever it is available for MS patients.