PatientsLikeMe/Biogen Study Evaluates Wearable Devices To Monitors Activity Of People With MS
Written by |

Cambridge, Massachusetts based PatientsLikeMe has released results of a novel study conducted in conjunction with pharmaceutical maker Biogen that demonstrated how people living with multiple sclerosis (MS) can employ wearable activity tracking devices to monitor, collect, and share their personal mobility data. That information can potentially be relevant for their clinicians, and for other MS patients.
Findings of the study are to be presented at the 67th American Academy of Neurology’s (AAN) Annual Meeting which is being in Washington, DC from April 18 to 25.
“MS impairs the ability to walk for many people with MS, yet we only assess walking ability in the limited time a patient is in the doctors office,” says Richard Rudick, MD, vice president for Value Based Medicine at Biogen. “Consumer devices can measure number of steps, distance walked, and sleep quality on a continuous basis in a persons home environment. These data could provide potentially important information to supplement office visit exams.”
The Biogen presentation “Remote Tracking of Walking Activity in MS patients in a Real World Setting” (poster P3.209) will be available during poster session III at the AAN Annual Meeting on Tuesday, April 21, 2015 at 5:00 p.m. ET
PatientsLikeMe and Biogen designed the study to assess the practicality and effectiveness of using a consumer wearable device to monitor activity among people with MS in real-world circumstances, by 248 PatientsLikeMe members recruited to participate and provided with Fitbit One activity trackers.
Information on patient demographics and levels of self-reported functional disability were obtained from the participant’s PatientsLikeMe profiles. The FitBit devices were mailed to participants with instructions on how to activate them and authorize data sharing between the manufacturer and PatientsLikeMe. Of the 248 study participants who received the devices, 213 (82%) activated the device with the Fitbit website and authorized PatientsLikeMe to access their data. Two-hundred three of those who authorized data sharing activated device synchronization the with FItBit service and produced tracking data. Participants synced an average 18.21 days of data over the course of the 21-day study (87% adherence).
As part of PatientsLikeMe’s member engagement framework, a live concierge service was available to participants to provide answers to technical and other questions. The study also took full advantage of the PatientsLikeMe platform and health tracking tools in order to engage participants with their data and with each other. Study participants could track their physical activity levels on the PatientsLikeMe website and connect with one another in the MS discussion forum to discuss changing symptoms, benefits and issues.
PatientsLikeMe Vice President of Innovation Paul Wicks, PhD observes that advances in wearable health technology have the potential to shed more light on disease characteristics. PatientsLikeMe is uniquely positioned to combine self-reported data with objective measurement and thereby help patients and researchers learn more from the data to impact self discovery and research.
[adrotate group=”4″]
“The three-week study had a lasting impact on its participants, who together took a total of 15 million steps and walked 6,820 miles, the distance from Boston to Beijing. I got positive reinforcement to do more each day,” comments PatientsLikeMe member and study participant Annette Smiling, who had never previously used a wearable activity tracker before, “and that really encouraged me. The Fitbit also allowed me to track what I was eating and how I was sleeping. I made more positive choices as a result.”
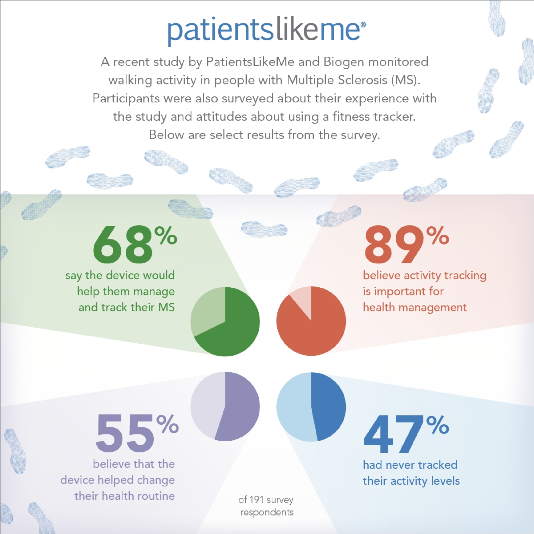
At the end of the study period, participants were surveyed to learn more about their experiences with the FitBit devices and about how they view the use of this technology for physical activity tracking. Of the 191 participants who responded to the post-study survey, 88 percent said the device was easy to use and to integrate into their daily routines; 83 percent affirmed intent to continue using the device after the study; and 68 percent believed that the device would be useful for helping them manage their MS.
PatientsLikeMe was co-founded in 2004 by three MIT engineers: brothers Benjamin and James Heywood and longtime friend Jeff Cole. Five years earlier, a third Heywood brother and friend of Mr. Cole’s, Stephen Heywood, was diagnosed with ALS (Lou Gehrig’s disease) at the age of 29. The Heywood family conducted a worldwide search fornformation and ideas that would extend and improve Stephen’s life. Inspired by Stephen Heywood’s experiences, the co-founders and team conceptualized and built a health data-sharing platform that it believes can transform the way patients manage their own conditions, change the way industry conducts research and improve patient care.
Today, James Heywood is chairman of PatientsLikeMe and was Founder and Former CEO of the ALS Therapy Development Institute. Ben Heywood heads up product strategy and the UX and engineering teams for the PatientsLike Mesite, and is also Chief Privacy Officer and overseer of PatientLikeMe Openness Philosophy.
Jeff Cole has been developing software for web applications for over 10 years, some of which time he’s spent helping out at the ALS Therapy Development Institute.
With patient network of more than 38,000 members, PatientsLikeMe’s MS community is the largest and most active MS research community online. Through the network, patients can connect with others experiencing the same symptoms and condition and track and share their own experiences. In the process, members generate data about the real-world nature of the disease that can help researchers, pharmaceutical companies, regulators, providers, and nonprofits develop more effective products, services and care for MS patients. With more than 325,000 total members, PatientsLikeMe is a trusted source for real-world disease information and a clinically robust resource that has published more than 60 peer-reviewed research studies.
For more information, visit:
https://www.patientslikeme.com


Leave a comment
Fill in the required fields to post. Your email address will not be published.