New MS Drug To Be Distributed By Global Supply Chain Distributor Durbin
Written by |
Medical Need Europe, a privately held pharmaceutical company headquartered in Sweden and focused on registration, distribution, marketing and sale of orphan drugs and niche speciality pharmaceuticals for treatment of rare diseases, has appointed London, U.K. based Durbin PLC, a leading global supplier and distributor of pharmaceuticals, to manage warehousing and supply chain for a range of Medical Need products.
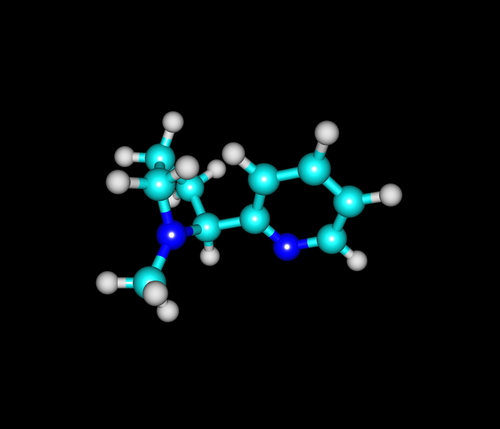
At the outset, this collaboration will involve a single product, Medical Need’s progressive multiple sclerosis (MS) management drug MD1003, which is currently under clinical development.
In April, Medical Need partner MedDay Pharmaceuticals, a biotechnology company focused on treatment of nervous system disorders, founded in 2011 by neurologist and neuroscientist Fredric Sedel, MD, PhD, and Guillaume Brion, announced positive results from the pivotal randomized 2:1, double-blinded, placebo-controlled Phase III clinical trial MS-SPI. The trial was conducted in 16 MS reference centers in France, and showed evidence of the efficacy and safety of MD1003, a highly concentrated pharmaceutical-grade biotin administered at a dose of 300 mg per day for treatment of primary and secondary progressive multiple sclerosis, a major area of unmet medical need. Treatment duration was one year.
The company reported that the MS-SPI clinical trial’s primary endpoint was met with a proportion of MS patients showing an improvement in EDSS (expanded disability status scale) or TW25 (timed 25-foot walk) at Month 9 and confirmed at Month 12 (p = 0.0051). The primary endpoint was supported by evidence of a substantial decrease in the risk of disease progression.
The patient population was defined as patients suffering from primary progressive Multiple Sclerosis (PPMS) or secondary progressive Multiple Sclerosis (SPMS) with EDSS progression within the two years prior to inclusion, and with EDSS ranging from 4.5 to 7. Patients with disease modifying therapy (DMT) introduced less than 3 months prior to inclusion; fampridine introduced less than 1 month prior to inclusion; or evidence of relapse or Gadolinium-MRI activity within the past year were excluded from the study.
The trial data were presented for the first time at The American Academy of Neurology (AAN) 2015 Annual Meeting’s Clinical Trials Plenary Session, at Washington DC. by the Principal Investigator, and the detailed data are expected to be published in a peer review journal in due course.
Durbin will coordinate distribution of MD1003 through its Managed Access Programme (MAP), which will enable patients with physician-assessed clinical need to receive the product before it is generally commercially available.
 “Initial feedback from neurologists and the MS community indicated that there will be requests for MD1003 on a Named Patient or Early Access basis,” explains Medical Need CEO Peder Walberg in a release. “We believe our collaboration with Durbin will strengthen our logistical setup and improve our ability to answer the requests for MD1003 in these markets.”
“Initial feedback from neurologists and the MS community indicated that there will be requests for MD1003 on a Named Patient or Early Access basis,” explains Medical Need CEO Peder Walberg in a release. “We believe our collaboration with Durbin will strengthen our logistical setup and improve our ability to answer the requests for MD1003 in these markets.”
“While we are very pleased with our existing partnership with BS Orphan, to provide medical support services for our products in the UK, Ireland and Netherlands,” Mr. Walberg continues, “our upcoming pipeline of products for unmet medical needs required further services within the current setup. Medical Need required an additional logistics partner with regulatory expertise and experience, which could be trusted to effectively manage the supply as requested and permitted. Durbin met these criteria, and we are very happy to be working with them.”
Established more than 50 years ago in 1963, and with over 20 years experience in Named Patient and Early Access programmes working with leading pharmaceutical and biotechnology companies to distribute products on a global scale, Durbin is one of the world’s leading specialist medical suppliers, sourcing and distributing pharmaceuticals, medical equipment and consumable supplies to healthcare professionals in over 180 countries.
Durbin’s main divisions encompass international sales, sales to NGOs and charities, clinical trials supply, managed access programs and imported unlicensed medicines to sexual health supplies, and storage and distribution of medical products and devices for third parties. The company has over 20,000 square feet warehouse facilities fitted out with a state-of-the-art evaporative cooling system and walk-in fridges for items requiring storage between 2-8C, located close to London’s Heathrow Airport, from which Durbin is able to supply anything from a single cold chain item to large-scale humanitarian projects. The company has preferential rates with all the major freight forwarding companies and can ship most items at a moment’s notice.
Durbin’s 100 strong multi-lingual workforce includes pharmacists, medical engineers and nurses, as well as commercial, warehouse and logistics staff possessing a wide range of in-house skills, and has set up its operation to act a one-stop-shop able to accommodate healthcare supply needs from local project level to national scale projects, supplying over 20,000 branded, generic, medical and consumable products. Durbin customers include multilateral organisations, NGOs, charities, religious health facilities, government agencies, pharmaceutical wholesalers and traders, hospitals, retail chemists/pharmacists and GPs.
Durbin has won several awards including International Business of the Year (Business Excellence Awards 2012), SME of the Year (Business Excellence Awards 2008) and the  Coutts Family Business Award (London/Greater London) in 2009. In 2013, Leslie Morgan, CEO of Durbin was awarded an Officer of the Most Excellent Order of the British Empire (OBE) in the Royal New Year’s Honours list for services to the Pharmaceutical Industry and to Charity, and was elected a Fellow of the Royal Pharmaceutical Society later that year.
Coutts Family Business Award (London/Greater London) in 2009. In 2013, Leslie Morgan, CEO of Durbin was awarded an Officer of the Most Excellent Order of the British Empire (OBE) in the Royal New Year’s Honours list for services to the Pharmaceutical Industry and to Charity, and was elected a Fellow of the Royal Pharmaceutical Society later that year.
Joining what was then the small retail pharmacy B&S Durbin Ltd. in 1976 as a pharmacist, Mr. Morgan began developing the export side of the business soon after as the company’s international sales director, traveling widely, sourcing medicines, and meeting new customers all over the world. In 1999, he bought the company, becoming Managing Director, and changed its name to Durbin PLC.
“Initially my goal was to find out what problems healthcare professionals were having in getting hold of medicines and supplies and to source whatever they needed as quickly and cost-effectively as possible,” said Mr. Morgan. “The whole operation still revolves around that philosophy and it works. Our customers call with a request and we find what they need and get it to them when they need it.”
Mr. Morgan’s philanthropic approach to business is very much part of the Durbin philosophy, and he spends a lot of time working with pharmaceutical and medical equipment organisations assisting them to donate in-date products to charities worldwide, his efforts thus far having resulted in over 20 million worth of donated stock.
 Commenting on the deal with Medical Need, Mr. Morgan observes; “Durbin has been managing several programs within the MS arena, and we look forward to continuing to contribute to improved patient care through access to MD1003 in these markets. A project of this scale requires a specialist global distributor with regulatory experience, and our previous work, along with patient focus positions us well for our partnership with Medical Need… a specialist and dynamic company which is dedicated to improving the lives of patients with unmet medical needs and we welcome the opportunity to work in partnership with both them and BS Orphan.”
Commenting on the deal with Medical Need, Mr. Morgan observes; “Durbin has been managing several programs within the MS arena, and we look forward to continuing to contribute to improved patient care through access to MD1003 in these markets. A project of this scale requires a specialist global distributor with regulatory experience, and our previous work, along with patient focus positions us well for our partnership with Medical Need… a specialist and dynamic company which is dedicated to improving the lives of patients with unmet medical needs and we welcome the opportunity to work in partnership with both them and BS Orphan.”
Further treatments for the management of patients with unmet clinical needs in gastrointestinal and other conditions are expected to launch under the Durbin-Medical Need distribution partnership in the coming months.
Sources:
Durbin PLC
Medical Need Europe
MedDay Pharmaceuticals
Image Credits:
Durbin PLC
Medical Need Europe
MedDay Pharmaceuticals


Nancy sullivan
When is the vaccine going to be available for all who need it?