Out on a Stim: The Pros and Cons of FES Devices (Part 1)
Written by |


First in a series.
For a minute or two, I felt like I was in a time machine.
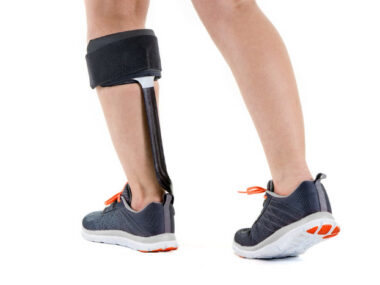
It was late March, and I was test driving (perhaps test walking) the WalkAide System, a functional electronic stimulation (FES) device designed, according to their website, “to improve walking ability of people living with foot drop.” Functional electronic stimulation, for the uninitiated, “applies small electrical pulses to paralyzed muscles to restore or improve their function.”
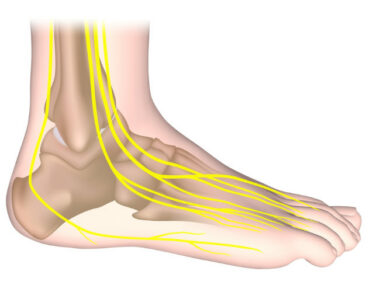
For foot-droppers like me, those pulses emanate from the FES device then travel along the peroneal nerve, which runs along the outside of legs and provides movement and sensation to the lower legs, feet, and toes. That stimulation and its results don’t work for everyone with MS, and the jury is still out (and has been out for a while) regarding the device’s long-term efficacy.
Still, on that day in March, the FES worked really well for me. Really well.
I’ve had foot drop for more than 10 years, and for a few months before FES, I’d used my walker to will myself around the block to try stemming muscle atrophy. Though I had used an AFO before, I no longer had the leg strength to stride through my gait properly while wearing it, and walking for any length of time had become very hard.
I pulled on my old running jacket and sweats, grabbed my cane, stepped out into our driveway, strapped the FES device onto my right calf, and then timidly turned it on. The pulse the device generates is triggered only by foot movement, and as I stood still, preparing to be pulsed, it did … nothing. Then I forced my foot to move and, sans cane, walked up my driveway.
By the end of the driveway, I was feeling good. I set the stopwatch app on my phone and walked around the block. With no walker, no cane, no assistive device or leaning against a telephone pole or fence. Nothing. Something between euphoria and delirium overtook me when I returned home and saw that I had made the trip in barely over five minutes. And while it didn’t burst the moment’s bubble, I noticed that I struggled to climb the two steps to my front door at the end of the walk.
Getting fitted for the device was pretty simple. First, I met with an orthotist at Hanger Clinic, who watched me walk and studied how my foot worked (or didn’t). Then he strapped the FES “cuff” around my right calf, just below my knee. Inside the cuff were two electrodes that HAD to be placed properly to make the correct contact against my leg and the peroneal nerve to work right. After that, the orthotist tweaked the device’s settings, reattached it to my leg, and then watched me walk again before handing me two packets of new electrodes and turning me loose.
I did lots of things with the device. I walked on the treadmill with it, played pool with it, walked all the way around the mall without resting, went to a crowded party and had drinks with it — everything that I could think of to test it. When I got the placement right, it improved my walking immensely, and again, it felt like I had turned back time by at least a few years. When I didn’t get the placement right, it was an “almost” improvement that helped me walk a little bit better yet still left me fearing that I would catch my toe before accidentally hitting the dirt.
About a year ago, I tried a similar device from Bioness, WalkAide’s main competitor in the U.S. At the time, they only offered an FES that was a foot-drop device with an electrode under the heel of the insole in the user’s shoe. The pulses travel up through the heel of the foot before stimulating the foot muscles, much as the WalkAide does. While the Bioness unit did work for me, I found its pulse “delivery system” disquieting.
Since then Bioness has released a new model, the L300 Go, which doesn’t require a heel piece. The investment for this kind of device is high enough that I feel compelled to try the new product before making any decision about them. I am scheduled to demo it on June 12. I’ll share more after that!
***
Note: Multiple Sclerosis News Today is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website. The opinions expressed in this column are not those of Multiple Sclerosis News Today or its parent company, Bionews Services, and are intended to spark discussion about issues pertaining to multiple sclerosis.

Dave
I use an FES from Odstock Medical in Salisbury, it is funded by the NHS..
I have used it now for 6 or 7 years, unfortunately it no longer works very well for me due to increased spasticity in my legs. It definately helped me to carry on being mobile for a few years, I would of been using a wheelchair much sooner without it. Don't know if the unit brought on my spasticity or it was natural desease progression, but it was definitely helpful for a few years.
dee middleton
They work great unfortunately very expensive and not covered by any insurance.
Wendy Vrana
I have been trying to get the walkaid, but insurance does not cover it. I have to have it for right and left side. I don't have $10,000 for them are things getting better for insurance covering them?
Mike Knight
Ugh. Unfortunately our insurance doesn't cover a penny for them, either. A reader on another piece told me to appeal insurance decisions multiple times if they refuse and to ask your PT or physiatrist to help (he said that approach worked for him). I think there are payment plans available (and if I/we buy one I'm sure that will be how we will do it). Thanks for reading my work, Wendy and all the best!
Wendy Vrana
Thank you so much for the information.
Mike Knight
Of course!
Daryl
I just purchased the Bioness GO PLUS system to replace my old L300 if you call Bioness they can give you some foundations that might help with cost, you just have to fill out some paperwork and pray for some help! We have tried a million times with the insurance, the just keep sending letters that said NO.
Rick Boehm
Have been considering a Bioness unit. Will wait until I hear your report on it. Did talk with my insurance and they will cover 80percent
Mike Knight
Good to hear from you, Rick, I hope you’re doing well! That’s great that your insurance will cover so much of it. Ive heard that ours might cover the electrodes (Walkaide’s last appx two weeks) which seems weird that they cover the supplies but not the device.
I’m cautiously looking forward to trying the Bioness unit now. The people at Hanger were very nice and let me really use their device long enough to thoroughly evaluate the unit, and that was so valuable (and something I'll ask of the Bionessfolks, too).
Rose Hesser
Hi Mike,
Through a bad experience Bioness is very backwards, first it took 2 weeks to meet with someone because they have one person doing all of NJ and some of PA. Then she let me try it for half hour and it felt fine and it worked pretty well, I asked if I could trial it and she said NO you need to purchase it at $5,530 plus tax and then you have 30 days to decide. If I opt to return the device within the first Thirty (30) days from the date of shipment, the total purchase price lesser a $595.00 usage fee will be refunded.
I at the time was excited (not about the money but because it worked well and I could do without my AFO brace. Hindsight is 20/20. After I got the brace, I had to meet with the rep so she could set it up, that took one week and a half. When she set it up it was working fine and then it was not working to my advantage I called her and asked if we could meet, she said the only time I can see you is 3 weeks from now, I said well then you need to give me an extension on my 30 days, they gave me an extra week. I had another issue and the same problem, but they would not extend it another time. I said OK I want my refund I went through "care credit". The response was " I am sorry the system didn’t work as planned.
Please be aware that by returning the Bioness L300 Go System all previous payments made toward the device will not apply to future purchases. The 30 day trial with the L300 Go System is for first time users and you will not have the option to trial the device in the future."
I thought that email was rude and someone that doesn't want business. I am not trying to sway you, I just wanted to share with you my experience.
Mike Knight
Thanks, Rose, this is really useful information. I believe I've seen other comments similar to yours, and my prior experience with Bioness was similar: their rep for Indiana covers several other states and it was very difficult getting time scheduled. It's good to have all of this background, because to your point, I think I/we have to go into these sorts of aids and support programs with our eyes wide open. Thank you for reading my column, I appreciate it and wish you the very best!
Julana
I tried the Bioness units a couple years ago. I needed the knee cuff and the thigh cuff. They worked great for me! Of course my insurance would not cover them... I ended up in the hospital with an exacerbation during my trial period and they were very willing to extend my time. I was scheduled for shoulder surgery as well Fter that so I opted to return until things settled down and maybe insurance would cover... I’m excited that they now have improved the unit!! I want to try again. The reps in Texas were great to work with!
Alberto Leonardi
Grazie Mike, ottimo articolo!
Mike Knight
Prego, Alberto!
Michelle Walsh
Hi Mike,
I was in the clinical trial for the Walkaide with Dr. Stein up here in Alberta,Canada years ago where this was initially invented. Over the years the device has strengthened my peroneal nerve or whatever causes the foot to drop to where I no longer have foot drop. Just wanted to give you my personal info on this device and the benefits of the FES. I hope you get the same positive results over time my fellow MSkuteer:)
Mike Knight
Thanks for the feedback and information, Michelle, and also for your support! I've wondered if the residual effect of wearing an FES would be positive, negative or negligible, so it's really valuable to me (and hopefully others of course) to hear how the FES has worked for you (and pretty cool that you were part of that trial, too). Thanks for reading the piece. And I'm kind of loving MSkuteer, too;)
Hattie
Mike, thank you for writing these articles.
I was given the Odstock FES device a few weeks ago and searched the internet for some hope, after having tried so hard to keep fit and well since diagnosis.
I saw Michelle's response and looked up the results from Dr Stein's trial. I found hope. So with much gratitute to you both, I am praying for my peroneal nerve to get stronger and sending you both warmest wishes.
Cindy
Hi, since you were part of a trial with very positive results does Alberta insurance cover any of the cost ? Thank U for your information
Kathy Allen
Hi I remember trying out the walk-aide in the office but not going ahead with it. I have had the Bioness for about 5 years. It definitely has helped me get further than I would without it. But recently I have been exercising with Trevor Wicken, MS Gym, & am finding myself rebalancing etc so I am walking again without it then using it when my foot drags again, experimenting.
I have no insurance coverage at all for Bioness. Since the company will not service the old product in two years , will buy new parts prior to end. Kathy
Mike Knight
Thanks so much for the information (and for reading this column), I appreciate it!
Kevin Lipetzky
Question for those who have drop foot in their right leg. Do you have trouble driving an automatic transmission vehicle? How did FES help your driving? Drop foot takes away the fun of driving.
Mike Knight
If I recall correctly, WalkAide recommends not using the FES while driving...and FWIW, drop foot does take a lot of fun out of driving to me.!
Samantha Jenne
I drive with my left foot. My vehicle was adapted for me. It’s wonderful.
Samantha
GARY SHAMBLEN
My insurance rejected my claim two times. For my third appeal my Neuro, in a Peer to Peer conference with my insurance co. Dr.declared this a medical necessity for me. They finally came up with 40% of cost for my FES. Bioness gave a discount for cash and I paid the balance. I have gained strength and nerve memory in my lower leg over the years.
Mike Knight
Thanks for the information, it's very helpful!
Kevin Lipetzky
Sounds like the companies selling FES want to remove all liabilities.
Pete
I've had the Bioness L300 Plus for 6 years. Like most, my insurance didn't cover any of the cost but I went forward with the purchase. It helped to extend my career by a few years before going on disability. To me it was worth it. The pros for purchasing: the stability it provided my left leg, helped to prevent foot drop plus increased my mobility. Even though my mobility has greatly decreased over the years, it is still better than w/o it. The cons: noises (beeps) it tends to make even while muted and once in awhile it misfires which has caused me to fall a couple of times. The residual effect of wearing this device has been negligible for me.
Bob P.
To all Who want the WALK Aide UNIT, the VA Hospital covers it if a Veteran! I'm inWilmington, DE.I hope this Helps!
Ed Tobias
Hi Mike,
I'm an L300 user (left foot drop) and test-walked the L300 Go last September. For me, the upgrade wasn't worth the cost. Here's my brief review. http://multiplesclerosisnewstoday.com/2017/09/26/ms-test-walking-the-new-bioness-l300-go/
Ed
Mike Knight
Hi Ed, and thanks so much for reading the column! I read your excellent review both when it came out and again last night. I'm looking forward to trying the L 300 go in a couple of weeks to see how it stacks up against your experience. Thank you so much for your help, I really appreciate it!
Sandra D
I have had PPMS for 12 years now and used the Walkaide for my job in neonatal nursing. It greatly assisted my left foot drop and kept me at work for 4 years before I had to resign because of fatigue. I used it for a few more years before my worsening fatigue/hip flexors became too weak and I stopped using it. I'm in Australia and my accountant was able to source a refund for me of around $1000 because I required it for my work. Originally the Walkaide cost around $5000 back in 2008. Expensive, but it helped keep me at work and mobile. Private health funds gave nothing back at the time.
Paul
How long did you use the AFO. I just got my AFO a few weeks ago and has made a significant difference in my daily walk. I also wonder how long before my leg weakens. But good for now. Thanks for the insight as it’s always good to hear from those who have been there.
Mike Knight
Thanks, Paul. I received the AFO in late April 2014 and, after I figured out how to incorporate it into my daily life (it's against the law where I live to wear that type of device while driving, had to figure out how to wear it comfortably during the day, etc.), it was super-useful for a couple of years following. In April 2016 I went on to long-term disability and while I did use the elliptical machine at the gym, went to a weekly Pilates for MS session and rode my bicycle routinely, I didn't walk as much thinking that I was generally staying in shape. In retrospect, I should have spent more time walking as now I think that those muscles/that system really needs routine and purposeful exercise. I am now embarking upon a focused fitness routine with hopes of recovering some strength in my legs to help reverse whatever I can, if I can. Thanks again for reading my column, I appreciate it and wish you the very best of luck!
John Cowburn
Hi Mike
I'm in the UK and the provision of FES devices are different from one Health Region to another, some will provide them whilst a neighboring Health Region may not!!!
My feet catch on the floor when I walk, one leg being worse than the other, but the main disability causing this is hip flexion rather than foot drop. I was tested for provision of an FES device, but due to hip flexion rather than foot drop, it made little if any difference and I was rejected for FES.
I think you are spot on about keeping on trying to walk as much as possible, unfortunately I have neglected this due to the fatigue produced and zero balance. My walking muscles definitely now show atrophy despite a lot of other exercise (weights, squats etc) So I am now determined to walk as much as possible (furthest is about 25 yards at a time) no matter how tiring. I am also trying to follow Trevor Wicken's "MS Gym" and the exercises you linked to in a previous post. John
Mike Knight
Thank you, John, I appreciate you taking the time to read my work! It sounds as if we are in very similar places in terms of condition. I, too, have avoided walking enough that the muscles in both legs have atrophied, though in my right (the one with foot drop) it is more significant of course. I think the combination of avoiding walking along with the effects of MS have created the perfect storm in terms of muscle strength. The question is whether I (we) can find the right approach for salvaging what's left. I have now incorporated walking with my rollator as part of my workout, and I do that for about five or six minutes every third day. It's not very pretty, but it has to be done! Not horribly long ago I was taught how to use the rollator (by walking closer to it, standing more erect and keeping both hymn breaks slightly depressed) and that was incredibly useful and makes the experience so much better. Thanks again for reading and for your input. The very best to you and keep me posted!
Marek Grabowski
I have just scheduled appointments for both WalkAide and Bioness! My insurance verbally claims that it would cover either of the devices 100%! I am in Denver, CO.
Thank you for bringing these to my attention.
Mike Knight
Thanks, Marek, that is great news (it sounds like you have very good insurance!) and I hope it works out well for you. Drop me a line and let me know how this works out for you if you would, please. Thanks for reading the column and your input, I appreciate it...
Andrea Pekarek
Hi Marek,
I am curious which unit—
Walk aide or Bioness did u decide to go with? I, too, am evaluating those two KAFO by OTTOBOCK StanceControl Free Walk.
I welcome any & all feedback & suggestions. Sincerely & Thank You
Andrea
Marc Falmer
What was your final decision? Did Walkaide or Bioness win?
mary ellen brubaker
I can afford and cannot get a site for either within 100 miles. no calls back, filled out on line forms, calls, emails. Are these people in business? Have been trying for two weeks.