Oligodendrocytes, Cells That Produce Myelin, Can Be Generated from Astrocytes, Study Reports
Written by |
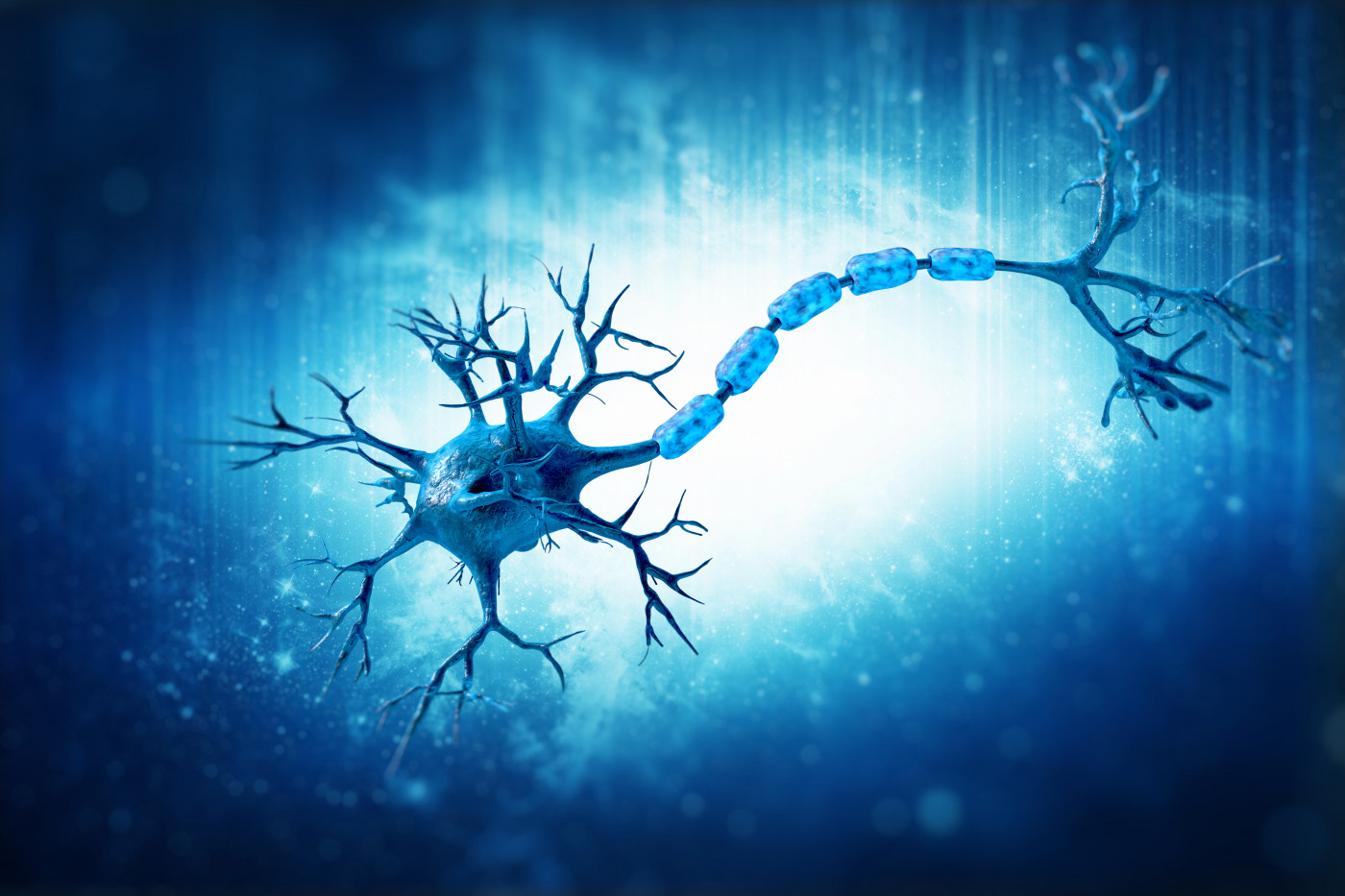
A molecule known as Sox10 enables brain cells called astrocytes to convert into myelin-forming oligodendrocytes, a new study in mice reports. The findings suggest an approach for myelin repair in patients with multiple sclerosis (MS) and similar disorders, its researchers said.
The study, “In vivo conversion of astrocytes into oligodendrocyte lineage cells with transcription factor Sox10; Promise for myelin repair in multiple sclerosis,” was published in the journal PLOS ONE.
Given the key role of oligodendrocyte pathology in MS, replacing these myelin-producing cells would be a promising therapeutic approach, particularly if autologous (of the same individual) sources of oligodendrocyte progenitor cells (OPCs) can be used.
Astrocytes – a cell involved in the formation of the blood-brain barrier and in response to injury – are the major component of glial scars, which develop after neural cell loss caused by degenerative diseases and traumatic injuries.
Prior research has shown that astrocytes may be converted into neurons (nerve cells) and stem-like cells in vitro by forcing the production of transcription factors, or molecules that control gene activity. As a result, changing astrocytes into progenitor cells has been attempted in vivo — inside a living organism — to enable tissue repair.
Both astrocytes and oligodendrocytes are types of glial cells. Astrocytes have been converted into myelinating cells with a micro RNA – tiny RNA molecules that also regulate gene activity – and a transcription factor not specific to oligodendrocytes. This inspired a research team at the Royan Institute for Stem Cell Biology and Technology in Iran and the University of California, Davis, to look for a transcription factor specific to oligodendrocytes that also showed an ability to change astrocytes into OPCs in vivo.
Discuss the latest research in the MS News Today forums!
Sox10 is a transcription factor produced constantly throughout oligodendrocyte progenitor cell development into mature oligodendrocytes, and is associated with the activation of myelination genes. In mice, Sox 10 is known to contribute to the generation of OPCs from fibroblasts, the main cells of connective tissues.
To explore the potential of Sox10 to convert astrocytes to oligodendrocytes, the researchers injected modified, harmless viral particles containing Sox10 and a fluorescent tag (to enable detection) into the area that connects the two sides of the brain, called corpus callosum, in mice with experimentally induced demyelination.
Control animals injected with particles lacking Sox10 were also analyzed.
These mice had extensive demyelination and astrocyte reactivity – a sign of brain damage – in the corpus callosum after 12 weeks, as well as impaired spatial memory.
After three weeks, most cells of control animals showed viral particles in astrocytes, while mice injected with Sox10 showed viral particles preferentially in cells with markers of oligodendrocyte lineage.
The result with Sox-10-containing viral particles was confirmed in mice with an eight-week interval between brain injection and analysis.
Researchers then collected astrocytes from mouse pups, injected them with the viral particles in vitro (outside a living body, i.e., in a lab dish), and transplanted the astrocytes into demyelinated corpus callosum 48 hours later. After three weeks, cells of this brain area showed oligodendrocyte progenitor markers (namely PdgfRalpha and NG2) as well as mature oligodendrocyte markers (Mbp and Plp).
These findings were confirmed by administering astrocytes with Sox10-containing viruses in an oligodendrocyte progenitor culture medium. The induced cells subsequently expressed OPC markers and showed morphological changes.
“Ultimately, our proof-of-principle study may suggest the achievability of in vivo astrocyte conversion into oligodendrocyte-like cells using Sox10 gene delivery into the demyelinated lesions,” the researchers wrote.
“We hope this approach lead to effective myelin repair in patients suffering from myelination deficit,” they added.

Graham A
Please work ... quickly!
Tom Harrison
IS this similar to Endecee?
http://endece.com/endece-neurals-ndc-1308-induces-remyelination-pre-clinical-multiple-sclerosis-ms-study/
Any followup planned?
Jessica Sherman
I would be interested in this therapy. I have RRMS on meds and I am relapsing now. I am 28 years old and still working. Now it’s effecting my hearing. I am so frustrated and tired of taking MS drugs that don’t work. Please advise.
Patrick Arieno
Please help me very soon!
wadalbertb
Can you learn more about the "oligodendrocyte pathology" that I am reading about in Oligodendrocytes, Cells That Produce Myelin, Can Be Generated from Astrocytes, Study Reports