FAQs about Ampyra
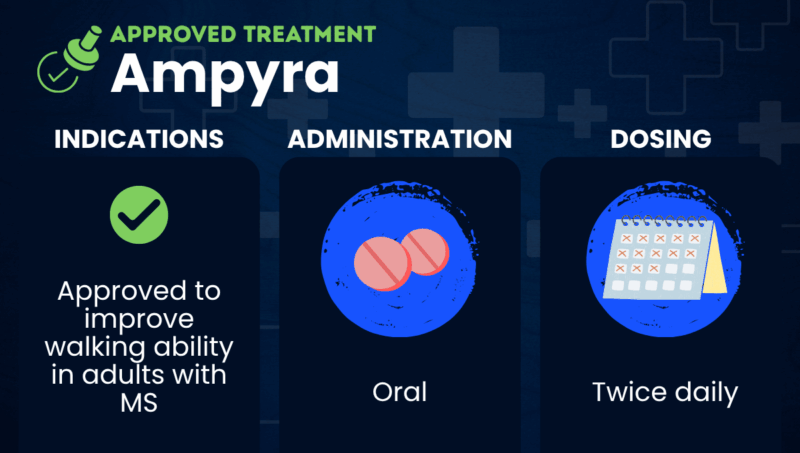
Ampyra was approved by the U.S. Food and Drug Administration in January 2010. It is indicated for easing walking difficulties associated with multiple sclerosis.
Ampyra has not been well-studied in pregnant people, but data from animal studies suggest it can cause harm to a developing fetus. Patients who become or plan to become pregnant while on the medication should discuss this issue with their healthcare provider.
Alcohol is not known to directly interact with Ampyra, but it may worsen certain side effects of the medication or exacerbate multiple sclerosis symptoms. Patients should consult with their healthcare team about the use of alcohol or other recreational drugs in combination with their treatments.
There have been reports of patients experiencing sustained improvements in walking speed within two months of treatment with Ampyra. However, not all people will experience improvements with this treatment, so patients should ask their doctor what to expect in their specific situation.
There have been no reports of hair loss or weight gain as side effects of Ampyra in clinical trials. Patients who experience unexpected reactions after starting on a new medication are advised to seek guidance from their healthcare team.
 Fact-checked by
Fact-checked by