FAQs about Mavenclad
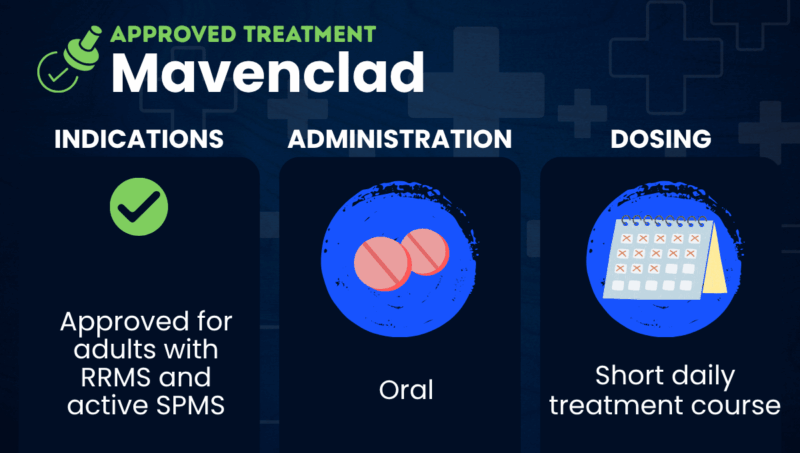
Mavenclad was approved by the U.S. Food and Drug Administration in March 2019 to treat relapsing forms of multiple sclerosis (MS), including relapsing-remitting MS and active secondary progressive MS.
Because it interferes with DNA replication, Mavenclad may cause damage to a developing fetus. Patients who have the potential to become pregnant should use effective contraception during dosing and for at least six months after their last dose.
Mavenclad is not known to interact with alcohol. However, both Mavenclad and alcohol can affect the liver, so drinking while on treatment may increase the risk of liver damage. Some side effects, such as headache, may also be more likely. Patients are advised to speak with their healthcare teams about whether it’s safe to consume alcohol during treatment.
In the CLARITY trial, which tested Mavenclad in more than 1,300 people with relapsing forms of multiple sclerosis, some benefits, such as fewer brain lesions, became evident as early as six months after starting treatment. However, responses can vary from person to person. Patients should talk with their healthcare team about what to expect based on their specific situation.
Weight gain has not been reported as a side effect of Mavenclad in clinical trials, but some people did experience hair loss. Individuals who experience unanticipated effects after starting treatment are advised to talk about such issues with their healthcare providers.
 Fact-checked by
Fact-checked by