Data From Multiple Sclerosis Study of Trimesta For Disability, Cognitive Improvements in Women To Be Presented at ACTRIMS-ECTRIMS 2014
Written by |
 Synthetic Biologics, Inc., a biotechnology company developing novel drug candidates targeting specific pathogens that cause serious infections and diseases, announced that its latest data on Trimesta™, regarding cognitive and disability measures in women suffering from multiple sclerosis (MS), will be presented at the 2014 Joint Americas and European Committees for Treatment and Research in Multiple Sclerosis Meeting (ACTRIMS-ECTRIMS) in Boston.
Synthetic Biologics, Inc., a biotechnology company developing novel drug candidates targeting specific pathogens that cause serious infections and diseases, announced that its latest data on Trimesta™, regarding cognitive and disability measures in women suffering from multiple sclerosis (MS), will be presented at the 2014 Joint Americas and European Committees for Treatment and Research in Multiple Sclerosis Meeting (ACTRIMS-ECTRIMS) in Boston.
Trimesta™ (oral estriol) is an investigational drug being evaluated in combination with Copaxone® (glatiramer acetate injection) in a randomized, double-blind, placebo-controlled Phase II clinical trial for the treatment of relapsing-remitting MS in women. A team of researchers led by Rhonda Voskuhl, M.D., Professor, Department of Neurology, and Jack H. Skirball,Chair in Multiple Sclerosis Research and Director, both part of the Multiple Sclerosis Program at University of California, Los Angeles (UCLA) School of Medicine, alongside investigators from 15 other U.S. centers, administered either Trimesta (8 milligrams orally per day) plus Copaxone® (20 milligrams per day), or placebo plus Copaxone® to patients randomized in this trial.
“We are thrilled that Dr. Voskuhl will present additional clinical findings for Trimesta, an oral agent for treating MS in women. The presentation provides further data on the Phase II topline efficacy results reported in April 2014 and is expected to provide details about the significant clinical outcome measures including relapse rate, disability, and cognition measures evaluated in this compelling trial,” stated Jeffrey Riley, Chief Executive Officer of Synthetic Biologics in a press release. “If the findings are supported in further studies, Trimesta, either alone or in combination with other MS drugs, could become a leading front-line therapy for women with MS due to its unique and differentiated anti-inflammatory, as well as neuroprotective benefits.”
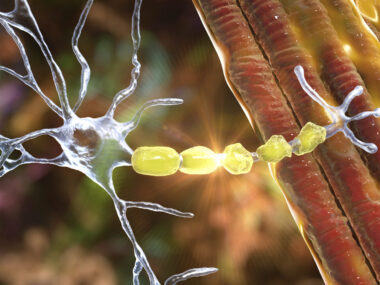
Multiple Sclerosis, an immune-mediated disease whereby progressive neurological degeneration leads to pain, loss of muscle control, paralysis, cognitive impairment and in some cases death, affects more than 2.3 million people worldwide, with approximately 300,000 to 400,000 patients diagnosed in the U.S. alone, the majority of which are women (65%). The annual worldwide sales of current MS therapies are estimated at $14.1 billion.
[adrotate group=”4″]
“The clinically significant improvement in cognitive testing in women taking Trimesta in this Phase II study is of high importance for MS specialists and patients. We believe the differentiated mechanism of action observed with Trimesta presents a new oral opportunity and has the potential to be an ideal added oral therapy to address significant unmet needs for women affected by this progressive, debilitating condition,” said Dr. Voskuhl
This investigator-initiated clinical trial is being supported by grants from the National Multiple Sclerosis Society (NMSS) together with the NMSS’s Southern California chapter, and the National Institutes of Health.

Leave a comment
Fill in the required fields to post. Your email address will not be published.