UPDATE: Australian Authorities Warn About Unapproved MS Stem Cell Treatments After Death in Russia
Written by |
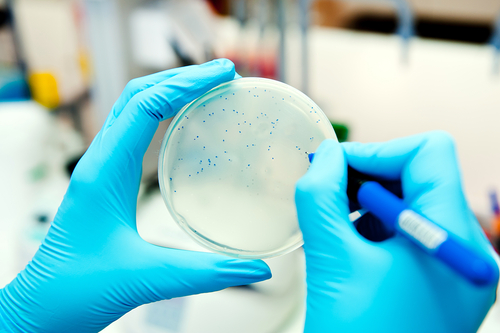
 Australian authorities are alerting Multiple Sclerosis patients to the risk of unproven stem cell treatments that are being performed both in the country and overseas, after the recent death of a woman in Russia undergoing a controversial treatment for the disease. The continued marketing campaigns to publicize stem cell tourism for patients with diseases like multiple sclerosis (MS) are raising several serious questions about the safety of both treatments and the clinics that offer them.
Australian authorities are alerting Multiple Sclerosis patients to the risk of unproven stem cell treatments that are being performed both in the country and overseas, after the recent death of a woman in Russia undergoing a controversial treatment for the disease. The continued marketing campaigns to publicize stem cell tourism for patients with diseases like multiple sclerosis (MS) are raising several serious questions about the safety of both treatments and the clinics that offer them.
Kellie van Meurs, an Australian mother of two, made headlines after she traveled to Moscow to undergo treatment for her rare neurological disorder, Stiff Person Syndrome. The woman died from a heart attack as a result of the treatment on July 19th. ”This sad case illustrates that some people are prepared to take the risk, but people should balance their high hopes with the acknowledgement that there is a risk,” said Stem Cells Australia’s head of education, ethics, law and community awareness, Megan Munsie.
Groups like the Adult Stem Cell Foundation often promote these trips, however, the treatments administered are untested and not accepted by mainstream science, according to the Australian authorities. Munsie believes that the combination of a proliferation of private clinics and the growth of consumers searching for alternative treatment options may be dangerous. ”It’s a commercial operation. We are concerned that patients don’t always see that there’s a conflict of interest. The person who is selling them a treatment is the person offering them advice,” she said.
The organization MS Australia has also alerted MS patients to the high risks of commonly searched treatments like autologous haematopoietic stem cell transplant (HSCT), the therapy that was administrated to Kellie van Meurs during her visit to Moscow. The treatment is pursued by MS patients and it involves the destruction of the patient’s immune system with high-dose chemotherapy and a subsequent “reboot” of the immune system using the patient’s own stem cells.
In order to avoid untested treatment options, Munsie advises that patients consult their physicians and take alternative treatment experiences for MS outlined in social media with a grain of salt. However, those with progressive forms of multiple sclerosis often become desperate for therapeutic options, since there are no FDA-approved treatments for MS past the preliminary relapsing-remitting form of the disease. In the U.S., those with progressive MS are treated with RRMS therapies, such as disease modifying or interferon-based drug, but these therapies are largely ineffective. In Australia, those with progressive MS often go completely untreated.
However, the National Health and Medical Research Council‘s (NHMRC) chief executive, Warwick Anderson, recommended that if patients want to enroll in unproven procedures, the best option is to sign up for a registered clinical trial. For example, in Australia, New Zealand-based Innate Immunotherapeutics’ is preparing to begin enrolling a clinical trial for its experimental drug MIS416 as a treatment for Secondary Progressive MS therapy.
According to an article on this subject recently posted on TheAge, there are currently more than 200 clinical trials analyzing the role of stem cells being conducted in the world, which is now thought to be able to help in the treatment of lung and kidney diseases, multiple sclerosis, autism, type 1 and 2 diabetes, prostate, and liver cancer.
Unproven and untested stem cell treatments may cause allergic reactions or rejection of the cells by the patient’s immune system and the development of cancer, both of which can be fatal. The only stem cell treatment currently deemed both safe and effective is blood stem cell transplantation to treat some blood and immune system disorders, according to the NHMRC.
The countries where patients often seek stem cell treatments are Russia, Thailand, India, China and the Bahamas. The executive director of Queensland-based Adult Stem Cell Foundation, Bruce Lahey, which deals with clinics in Shanghai, Bangkok and Moscow, said that the organization “supports HSCT, but we say it has limitations and it’s not for everybody. We’ve done the due diligence on the clinics we send our people to and we’ve never had one serious adverse event.”

Glenn Armstrong
I have MS and am in Moscow now going through the treatment. The hospital has performed this treatment over 400 times with one death unrelated to MS. It is proven - many times over. It offers MS sufferers hope where there is none otherwise. We sit and watch our bodies slowly become paralysed or we do something to stop it. More MUST be done to embrace this treatment for all MS sufferers as time is running out for so many. I believe it is negligent on behalf of the Aust Govt that this treatment is not opened up to all who desperately need it. Learn from the like of Moscow and use it as best practice. The time for more talk and ridiculous tails is over
Rich
Agreed, the glacial, litigation-bound pace of medical progress in the USA is a travesty. I know we need to be careful about risk, but making it illegal to pursue hopeful therapies except by taking the lawyer-approved clinical study route and not allowing people to choose their own level of safety is immoral and wrong. The balance has to shift away from a hideous fearful caution towards action to reflect the accelerating pace of medical technology.
Check out this recent report: http://multiplesclerosisnewstoday.com/2014/08/08/nuc-reports-promising-results-of-stem-cell-transplants-in-ms-patients/