A Step Toward Multiple Sclerosis Treatment? Phase 2 ANTI-LINGO-1 Results Announced
Written by |
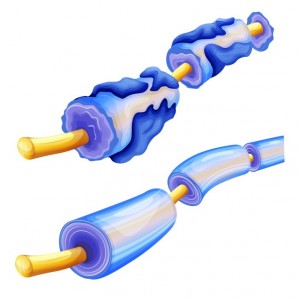 Biogen Idec, a Cambridge Massachusetts Biotechnology company, has released results from its Phase 2 acute optic neuritis (AON) RENEW trial. The trial tested anti-LINGO-1, a medication that restores myelin — a fatty substance that facilitates nerve cell impulses by wrapping around them and providing insulation. The trial results indicate that anti-LINGO-1 could help to repair the damaged visual system.
Biogen Idec, a Cambridge Massachusetts Biotechnology company, has released results from its Phase 2 acute optic neuritis (AON) RENEW trial. The trial tested anti-LINGO-1, a medication that restores myelin — a fatty substance that facilitates nerve cell impulses by wrapping around them and providing insulation. The trial results indicate that anti-LINGO-1 could help to repair the damaged visual system.
RENEW is one component of Biogen Idec’s program to develop anti-LINGO-1, which includes the SYNERGY multiple sclerosis trial. SYNERGY is an ongoing, separate Phase 2 study of anti-LINGO-1 in people with relapsing multiple sclerosis. Biogen Idec expects results for the SYNERGY trial in 2016.
In multiple sclerosis, the deterioration of myelin due to an autoimmune response accounts for loss of movement and other debilitating neurological symptoms. Targeting LINGO-1 might be a way to repair and increase myelin, as well as to help other nervous system cells to survive. LINGO-1 stands for: leucine-rich repeat and Ig domain-containing, Nogo receptor interacting protein. This protein is found in the central nervous system on two types of cells: oligodendrocytes; which provide myelin, and neurons; the information-processing cells of the nervous system. The normal function of LINGO-1 is to stop neurons and oligodendrocytes from further developing once the nervous system is fully formed. Inhibiting LINGO-1 could enable repair following injury.
AON is a disease of the visual system, in which loss of myelin sheath and neuron injury occur, causing damage to the optic nerve. Loss of vision can result. Because myelin loss in AON is well-understood and in one area of the body, studying anti-LINGO-1 in this disease is considered a good first step to test if the drug works. The next step will be to study multiple sclerosis, which is a more complex disease.
Results of the RENEW trial showed that giving patients anti-LINGO-1 improved standard measurements of vision and eye function by 24 weeks following the start of medication, compared to a group that received a placebo. Eighty-two individuals were included in this study.
“We believe the RENEW results are encouraging, as this is the first clinical trial to provide evidence of biological repair in the central nervous system (CNS) by facilitating remyelination following an acute inflammatory injury,” remarked Alfred Sandrock, M.D., Ph.D., group senior vice president and chief medical officer at Biogen Idec. “We look forward to the SYNERGY results in 2016 to further advance our understanding of this molecule in MS, including a full dose response. The totality of the data from the two Phase 2 studies may provide us with a clearer understanding of anti-LINGO-1’s clinical potential.”
Further data from the SYNERGY trial will shed light on whether anti-LINGO-1 can restore myelin and reduce symptoms in multiple sclerosis, providing more progress toward a possible treatment.



victor manuel chao
i would like more information as soon you know if it is working,thanks
Sophie Seesing
Dealing with SPMS, would like to join in testing the anti Lingo medicin.
Thanks for all the good work..
Best regards,
Sophie
Alisa Woods
The next trial results will be from SYNERGY and should be available in 2016.
https://www.nationalmssociety.org/Chapters/AZA/Calendar/Programs/Synergy-%28Lingo-1%29-Clinical-Trial
Kristi Koppmann
Will Anti-Lingo be available on 2016? I have been battling this monster for 40 years. IT IS TIME TO PUT IT DOWN!!!
adriana
I'd like to get into the clinical trial of Lingo-1
but I can't find the way to get into
can you help me or suggest somethine
many thanks
Adriana
https://www.biogen.com/en_us/research-pipeline/clinical-trials/how-to-participate.html
Peter
tiež by ma zaujímali vysledky z Lingo-1 taktiež by som sa odhodlal skusit liečbu - môžete mi prosím posielať info. ako pokračuje vyskum-Dakujem veľmi pekne.
Biogen Idec runs this program:
https://www.biogen.com/en_us/research-pipeline/clinical-trials/how-to-participate.html
fadi samaan
I would like to ask you my wife has been illness since 2000 until now so the attacks came to her body every three years but after she had a babies in 2010 so she didn't exposed an attac until now thanks.
John Grewe
I would like to be a part of an anti-LINGO-1 trial.
I'm in a wheelchair now, currently on Tecfidera and long to lay this disease to rest.
Biogen Idec runs this program, here is the link for potential participants:
https://www.biogen.com/en_us/research-pipeline/clinical-trials/how-to-participate.html
sandra
Hi this is Sandra I tried that link but it says page is not found
Try emailing:
[email protected]
linda kilcollins
Please, want to get on study. thanks
Linda Kilcollins 414 788-7317
Also their email for inquiries about participation:
[email protected]
Peter Reištetter
I am on the course and outcome of disease treatment Ling SM-1 comes from Slovakia and I, who have 30 years on sick SM'm walking, but I would like to know if it is possible the formation of myelin in such long-term illness? Among the Subscriber uses Copaxone eight years before that I did not use anything, because I did not meet the conditions of use interferon or immunomodulators = two attacks per year or three attacks in two years, I met after 21 years of disease that I "I had two attacks a year . I have RRMS. I'll be glad when I can treat the results LINGO-1 informed. Sincerely Reištetter Peter.
sandra
I've tried that link however, it's says it's not available
maxine semanchyk
I would love to get into the trials for anti lingo 1.....
Lisa Murphy
** If you want to participate in trials...**
If you are in the US or abroad, the website "www.clinical trials.gov" lists all clinical trials with US sponsors. (The US government requires sponsors to complete these listings.)
In the search box, enter "Anti-lingo 1" and the relevant studies will come up. Click on the various studies to see what interests you. If a study is actively enrolling patients and you would like to participate, review the inclusion and exclusion criteria to see if you are a match. If you are a match, scroll to the bottom of the page where the participating investigative clinical sites/centers are to see if there is a hospital or clinic/ medical center in your area that is participating. If there is one, the contact information for the center should be provided on this website.
This is the best way to find out what clinical trials are available to patients, and where.
For a broader search for other trials, you could search under "multiple sclerosis" or some other investigational drug's name.
Best wishes!