Mylan Confirms Continued Patent Proceedings Concerning MS Therapy Copaxone
Written by |
According to a press release from Mylan N.V., the U.S. Patent and Trademark Office (PTO) has issued an inter partes review (IPR) proceeding on all claims against a third Copaxone patent for the 40 mg/mL (U.S. Patent No. 8,969,302) formulation by Yeda Research & Development Co., Ltd. and licensed to Teva Pharmaceuticals Industries Ltd. The news represents continued patent challenges and efforts by other pharmaceutical companies to produce generic forms of Copaxone, which is a leading multiple sclerosis therapy for the relapsing-remitting form of the disease.
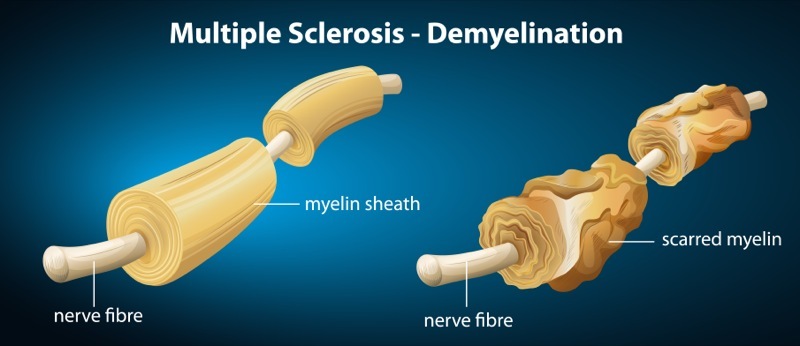
Copaxone is delivered via subcutaneous injection and has been found to be effective for many patients with RRMS in mitigating the number and severity of relapses. Multiple Sclerosis occurs when the nerve cells present a different morphology, namely, when the insulating covers of myelin around the cells and spinal cord are damaged, leading to the disruption of the ability of communication among the different parts of the nervous system. Due to this atypical morphology, the immune system identifies those cells as a potential target to eliminate by releasing biological markers, attacking the central nervous system (CNS), which includes brain, spinal cord and optic nerves. All in all, MS results from an immune-mediated process. While there are different forms of the disease, the relapsing-remitting form is the most common, making therapies such as Copaxone widely prescribed to control such exacerbations.
Copaxone is composed of Glatiramer Acetate (GA) , a polymer with immune response regulation properties. Once in the body, the GA polymer competes against the biological markers to bind to the myelin fibers. In this way, the immune reaction against the CNS becomes less intense.
RELATED: Mitochondria May Play a Role in MS Development and Progression
Last year, the U.S. Food and Drug Administration (FDA) accepted a new drug application (ANDA) filing for three-times-per-week Glatiramer Acetate Injection 40 mg/mL, developed by Mylan, a generic version of Copaxone 40 mg/mL. Mylan believes that their generic version of the drug shows promise for being granted 180 days of marketing exclusivity in the US after FDA approval. The expiration of the original Teva patent for Copaxone has spurred on patent challenges and legal action as drug manufacturers move to offer generic versions of the therapy.
In this latest development, the PTO has instituted IPR proceedings against two related Copaxone® 40 mg/mL patents, the U.S. Patent Nos. 8,232,250 and 8,399,413 on all claims, and the oral hearing will take place in May 2016.
Mylan is one of the world’s leading generic and specialty pharmaceutical company, with its headquarters in the United Kingdom. It has around 1,400 products in its portfolio.



Shirley Webber
Is there anything new for spms or anything being researched. THANK YOU