Foralumab reduces microglial activity, inflammation in nonactive SPMS
Nasal spray therapy also increased levels of neuroprotective proteins
Written by |
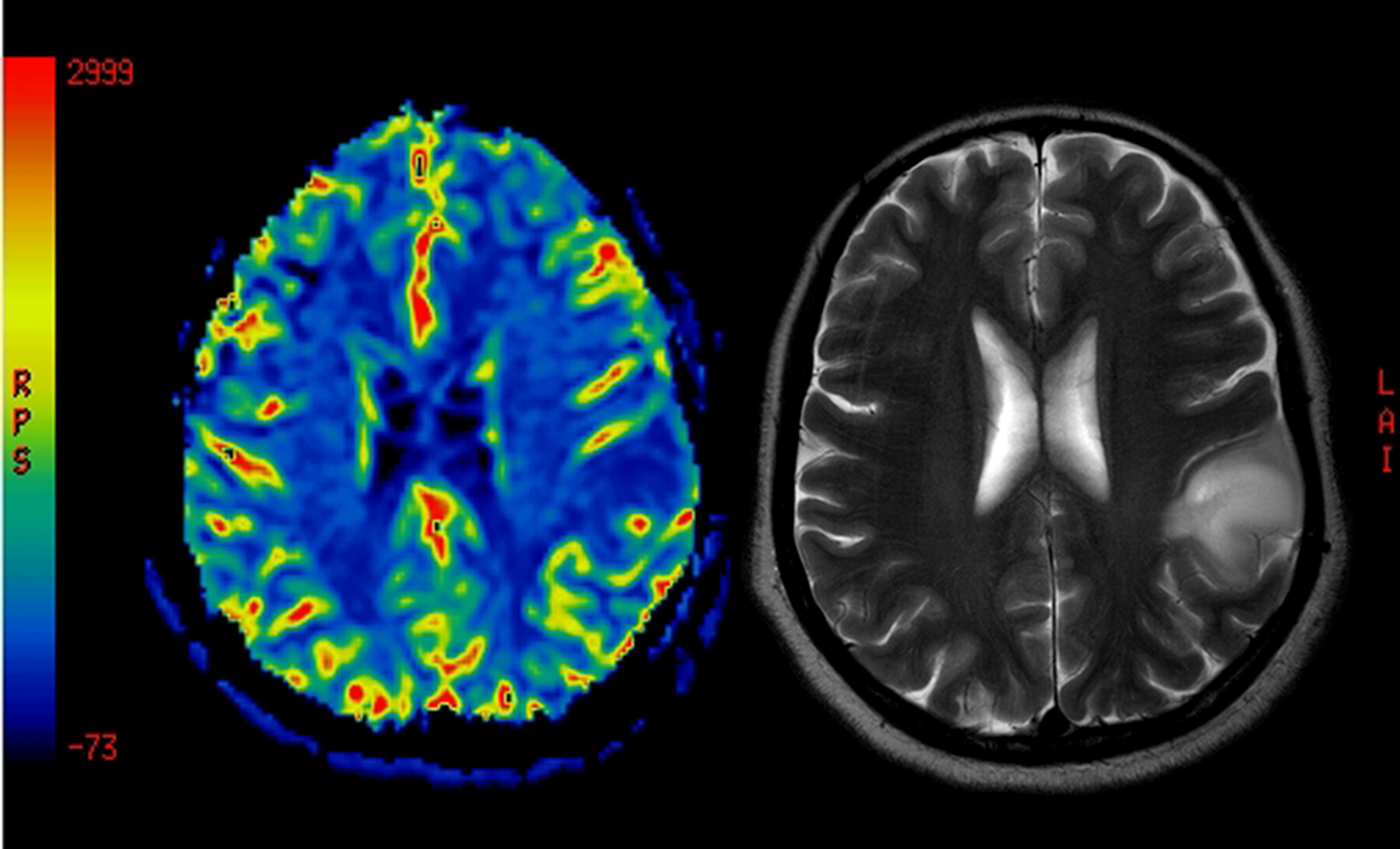
An image of brain scan.
- Nasal spray foralumab shows promise for nonactive secondary progressive multiple sclerosis.
- It reduces brain inflammation and microglial activity and increases neuroprotective proteins.
- Early studies showed stable disability and reduced fatigue; a Phase 2a trial is ongoing.
Treatment with the experimental nasal spray therapy foralumab reduced microglial activity in people with nonactive secondary progressive multiple sclerosis (SPMS), according to new data from an expanded access program.
Microglia, the brain’s resident immune cells, are believed to contribute to the chronic inflammatory processes that drive gradual disease progression in the absence of relapses. These reductions in microglial activity were significantly associated with decreased markers of inflammation in the spinal fluid and increased levels of neuroprotective proteins.
A team led by scientists at Brigham and Women’s Hospital in Boston presented the findings in a poster titled, “Nasal foralumab downregulates CSF inflammation and upregulates CSF neuroprotective proteomic pathways which correlate with [F-18]PBR06-PET imaging in na-SPMS with PIRA.”
“Nasal foralumab continues to show a unique ability to dampen smoldering [brain and spinal cord] inflammation while promoting neuroprotection,” Howard L. Weiner, MD, co-author of the study at Brigham and Women’s, said in a press release from Tiziana Life Sciences, the company developing foralumab.
Foralumab targets key protein on immune T-cells
Multiple sclerosis (MS) is driven by inflammation that damages healthy tissue in the brain and spinal cord. The most common form of MS is relapsing-remitting MS (RRMS), which is characterized by relapses in which symptoms suddenly worsen, followed by periods of remission where symptoms ease. Over time, some people with RRMS will progress to SPMS, which is marked by symptoms that gradually worsen over time, independent of relapses.
Some people with SPMS continue to experience relapses, and many of the MS therapies approved for RRMS can be used in these patients. But available treatments are much more limited for those with nonactive SPMS disease, who do not experience relapses or show signs of acute inflammation on MRI scans.
Foralumab is an antibody therapy that works by targeting the CD3 protein on immune T-cells. This is expected to activate regulatory T-cells, which help calm excessive immune responses, while suppressing the pro-inflammatory T-cells that cause damage in MS. The therapy has also been shown to reduce the activity of microglia, which are believed to drive disease progression in nonactive SPMS.
An expanded access program tested foralumab in 10 people with nonactive SPMS, most of whom had been experiencing worsening disability despite treatment. After at least six months of treatment, disability levels remained stable or were slightly reduced — indicating an easing of symptoms — and most patients experienced clinically meaningful reductions in fatigue.
Measures of walking ability, hand dexterity, cognition, and visual function also remained largely stable.
Phase 2a trial further evaluating experimental therapy
In the new analysis, researchers examined microglial inflammatory activity using the PET imaging tracer [18F]PBR06. Across the 10 patients, a total of 14 scans were collected before and after treatment.
Results showed that [18F]PBR06-PET scores decreased significantly with foralumab, indicating that the nasal therapy dampened the activity of microglia. A significant reduction in PET signals was observed as early as three months after treatment initiation, and levels continued to decline through the last follow-up assessment at a median of 7.5 months.
Analyses of spinal fluid done in parallel with the PET scans also suggested a reduction in microglial activity. Data indicated that foralumab decreased markers of inflammation, such as IFNAR1 and LY86, while increasing neuroprotective proteins such as MEG10.
This late-breaking poster provides the first direct link between reduced microglial PET signal and favorable [spinal fluid protein] shifts during nasal foralumab treatment in [nonactive SPMS.
These changes showed strong correlations with PET imaging scores, meaning that higher reductions in [18F]PBR06-PET signals were significantly associated with greater decreases in inflammatory markers and increases in neuroprotective proteins.
“This late-breaking poster provides the first direct link between reduced microglial PET signal and favorable [spinal fluid protein] shifts during nasal foralumab treatment in [nonactive SPMS],” said Tarun Singhal, MD, lead author and neurologist at Brigham and Women’s Hospital. “The correlations demonstrate that [F-18]PBR06-PET is biologically tied to the inflammatory and neurodegenerative processes driving progression in SPMS, and that [spinal fluid proteins] can serve as a practical biomarker of therapeutic response.”
Tiziana is now running a Phase 2a clinical trial (NCT06292923) to further evaluate the experimental therapy. The trial aims to enroll about 54 adults with nonactive SPMS who have experienced continued accumulation of disability despite at least two years of treatment with available MS therapies. Recruitment is ongoing at seven sites in the U.S.
Participants in the Phase 2a study will be randomly assigned to receive foralumab daily at one of two doses or a placebo for about three months. The study’s main goals are to assess the safety of the experimental therapy, its effect on nasal symptoms, and changes in microglial activity on [18F]PBR06-PET scans.