MS Progression Halted Using Stem Cells Derived from Gum Tissue
Written by |
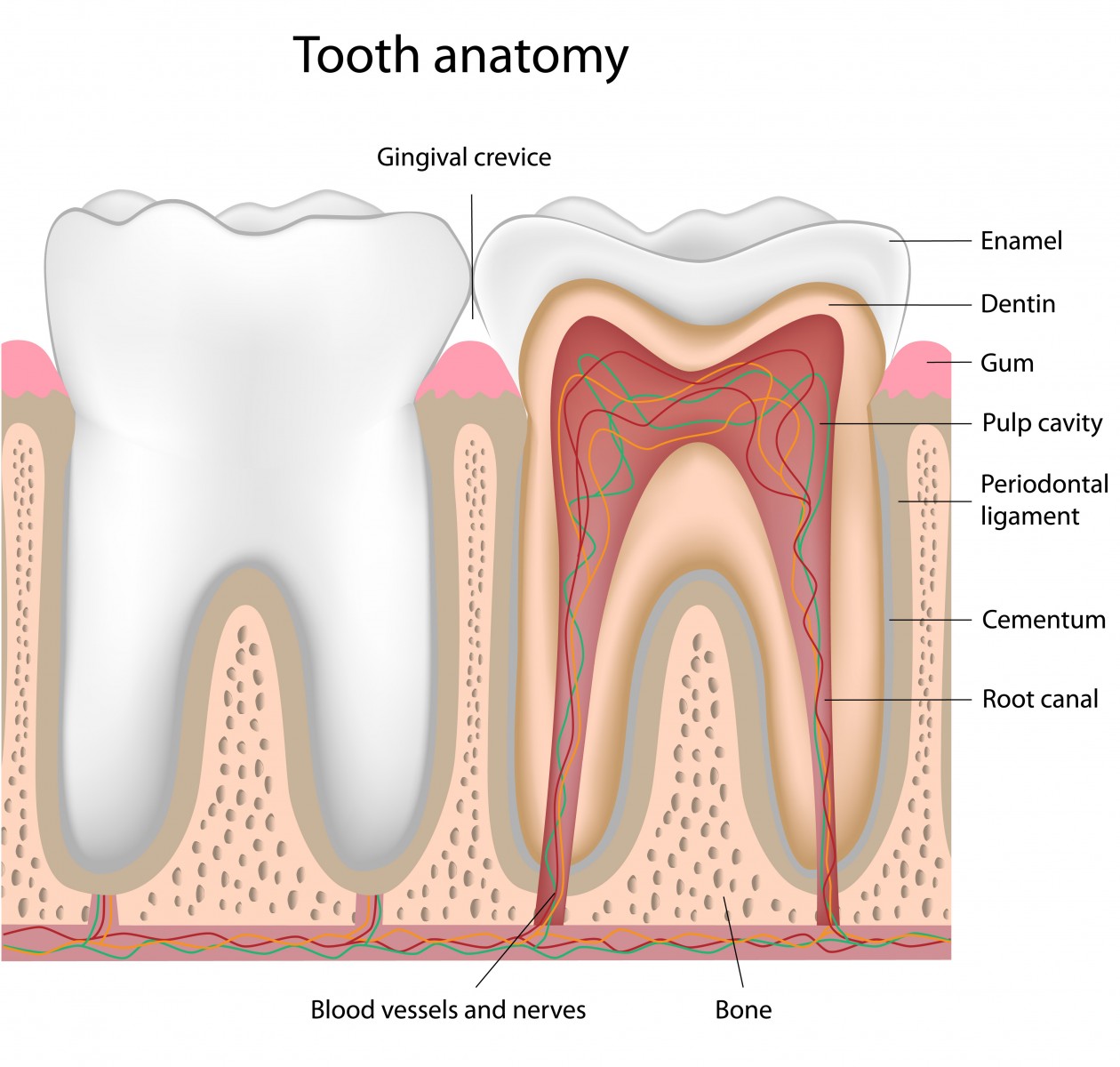
A new source of stem cells, the mesenchymal stem cells (MSCs) isolated from the human periodontal ligament (hPDLSCs), offers a potential treatment capable of halting multiple sclerosis (MS) progression. The study, “Alternative source of stem cells derived from human periodontal ligament: a new treatment for experimental autoimmune encephalomyelitis,” was published in the Stem Cell Research & Therapy journal.
Current treatments for MS are administered only as a palliative relief, without a cure, and many are associated with adverse side effects. The development of techniques that allow for harvesting and culture of MSCs, especially those derived from the bone marrow, aids investigations into its properties in promoting tissue repair and disease control in the established mouse model for human MS, the experimental autoimmune encephalomyelitis (EAE) model.
MSCs are known to have the potential to differentiate into several types of central nervous system (CNS) cells and the capacity to modulate the immune system at CNS lesions, enhancing the remyelination and repairing processes. However, the mechanisms regulating these processes remain ill-defined.
The Future of Stem Cell Therapy for Multiple Sclerosis Patients Relies on Clinical Trial Participation
A research team investigated the therapeutic potential for MS of an alternative source of stem cells that could be less invasive for removal. They chose the MSCs derived from the oral cavity, specifically the hPDLSCs. These cells are isolated from the periodontium, a tissue that supports the teeth in their sockets and allows mastication.
Researchers induced EAE in mice by injecting myelin oligodendroglial glycoprotein peptide (MOG). Mice were followed for 48 hours after injection for signs of EAE and weight loss. Within 14 days after immunization (at the period of disease onset), the EAE mice were administered a single intravenous injection of hPDLSCs. After an incubation period of 56 days, the mice’s spinal cord and brain tissues were analyzed.
The analysis showed that treatment with hPDLSCs has potential neuroprotective effects against EAE, as researchers observed diminished clinical signs, and decreased lymphocytic infiltration and demyelination in the spinal cord and brain tissues. Administration of hPDLSCs also exerted a modulatory effect on the expression of several inflammatory cells (D4 and CD8α T cells) and inflammation key markers [tumor necrosis factor-α, interleukin (IL)-1β, IL-10, glial fibrillary acidic protein, Nrf2 and Foxp3].
In conclusion, the results suggest that transplantation of hPDLSCs may become a new therapeutic strategy for halting MS progression.


Silvy
Very interesting and good news!
You can find more information about stem cells in our "stem cells for patient"
FB group, https://www.facebook.com/groups/721343281305825/
Come and join us. :-)
Silvia
Rhonda
How wonderful if this is found to be a way to stop this disease!! Absolutely interested in something that has no potential for PML nor side effects. As I am sure all MSers are hoping and praying for the ONE!!
Stuart
GREAT news! - Need further studies