Ocrevus (ocrelizumab) for multiple sclerosis
What is Ocrevus for MS?
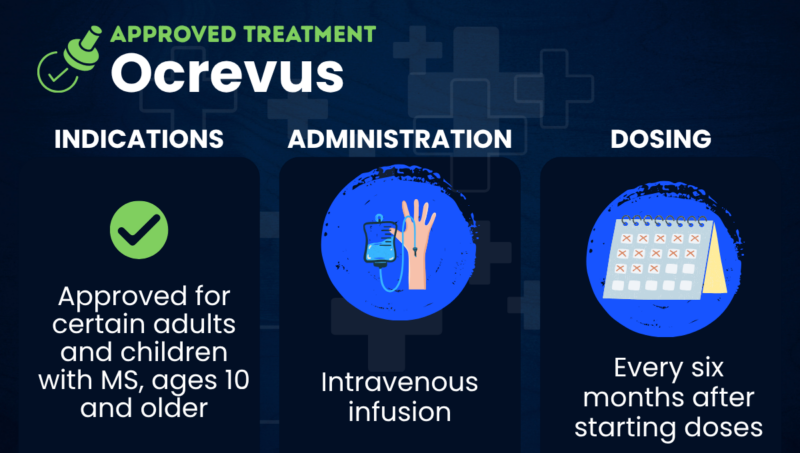
Ocrevus (ocrelizumab) is an approved infusion therapy that’s indicated for adults with relapsing forms of multiple sclerosis (MS), specifically, clinically isolated syndrome (CIS), relapsing-remitting MS (RRMS), and active secondary progressive MS (SPMS).
It is also approved for adults with primary progressive MS (PPMS) and for certain children, ages 1o and older, with RRMS.
Administered via an intravenous infusion, that is, directly into the bloodstream, the therapy is used to reduce the frequency of relapses, lower the number of lesions on MRI scans, and slow disability progression.
The active ingredient in Ocrevus is an antibody that targets the CD20 protein on the surface of B-cells, a group of immune cells implicated in the inflammatory attacks that damage the nervous system in MS. By lowering levels of these cells, Ocrevus is expected to ease MS-driving inflammation, helping to relieve symptoms and slow disease worsening.
Ocrevus is marketed by Genentech, a member of the Roche Group, which also developed a more convenient formulation of the medication, called Ocrevus Zunovo. The newer therapy is given via a 10-minute injection under the skin, known as a subcutaneous injection.
| Brand Name: | Ocrevus |
| Chemical Name: | Ocrelizumab |
| Usage: | Used to reduce disease activity and slow disability worsening in adults with relapsing forms of MS and PPMS, and in children with RRMS |
| Administration: | Intravenous infusion |
Who can take Ocrevus?
Ocrevus is approved in the U.S. to treat:
- adults with relapsing forms of MS, including CIS, RRMS, and active SPMS
- adults with PPMS
- children with RRMS, ages 10 and older, who weigh at least 25 kg (55 pounds)
It is contraindicated, or should not be used, in people who have:
- an active hepatitis B virus infection
- a history of a life-threatening infusion reaction to the medication
Ocrevus is also approved for adults with RRMS and PPMS in many other regions, including the European Union and Canada, although specific indications may vary.
How is Ocrevus administered?
Ocrevus is given via intravenous infusions that are administered by an experienced healthcare provider. The initial starting dose is split into two infusions, given two weeks apart, after which the medication is given every six months.
Dosing is weight-based as follows:
- patients weighing at least 35 kg (77 pounds): each starting dose contains 300 mg of the active ingredient; subsequent infusions have 600 mg
- children with weights less than 35 kg: each starting dose contains 150 mg; subsequent infusions have 300 mg
The infusions typically take 2-3.5 hours, but this can vary. Before each infusion, patients are premedicated with anti-inflammatory corticosteroids and antihistamines to prevent infusion reactions. All are monitored for at least an hour after the infusion.
Ocrevus in clinical trials
A few trials supported the approval of Ocrevus for its different indications. In all studies, Ocrevus was administered at its now-approved dosing regimen.
- Adults with relapsing forms of MS: A pair of similarly designed Phase 3 trials, OPERA I (NCT01247324) and OPERA II (NCT01412333), collectively enrolled 1,656 adults with relapsing MS, all of whom were randomly assigned to receive Ocrevus or Rebif (interferon beta-1a), an older MS medication, for about two years. In both trials, Ocrevus was associated with significant reductions in relapse rates compared with Rebif, as well as a lower risk of disability worsening and fewer brain lesions.
- Adults with PPMS: The Phase 3 ORATORIO trial (NCT01194570) enrolled 732 adults with PPMS, who were assigned to receive Ocrevus or a placebo for at least 120 weeks, or about 2.3 years. The results showed that Ocrevus significantly reduced the risk of disability progression, while also slowing declines in walking ability, preventing brain tissue loss, and lowering the number of new or enlarging brain lesions.
- Children with RRMS: The Phase 3 OPERETTA 2 trial (NCT05123703) enrolled 187 children and adolescents with RRMS, ages 10-17, who received either Ocrevus or Gilenya (fingolimod) — the only other MS therapy approved in the U.S. for children — for two years. Ocrevus reduced the risk of relapses and the proportion of patients with MRI activity compared with Gilenya, data showed.
Ocrevus side effects
The most common side effects associated with Ocrevus in adults and children include:
- upper and lower respiratory infections
- infusion reactions
- skin infections
Infusion reactions are more common after the first infusion and can become serious or life-threatening. Should they occur, the infusion may need to be paused, slowed, or stopped, depending on the severity.
Ocrevus also carries warnings for other potentially serious adverse events that require careful monitoring. These include:
- low antibody levels, which increases the risk of infections
- serious or fatal infections, including progressive multifocal leukoencephalopathy, a rare and life-threatening brain infection
- an increased risk of cancer, including breast cancer
- inflammation in the large intestine (colitis)
- liver injury
In some cases, these adverse events may require discontinuation of Ocrevus.
The effects of Ocrevus in pregnancy are not well established, but animal data suggest it could cause harm to a developing fetus. Female patients of reproductive potential are advised to use effective contraception while receiving Ocrevus and for six months after the last infusion.
Multiple Sclerosis News Today is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by 

