#ECTRIMS2016 – Stem Cell Transplantation Shows High Efficiency
Written by |
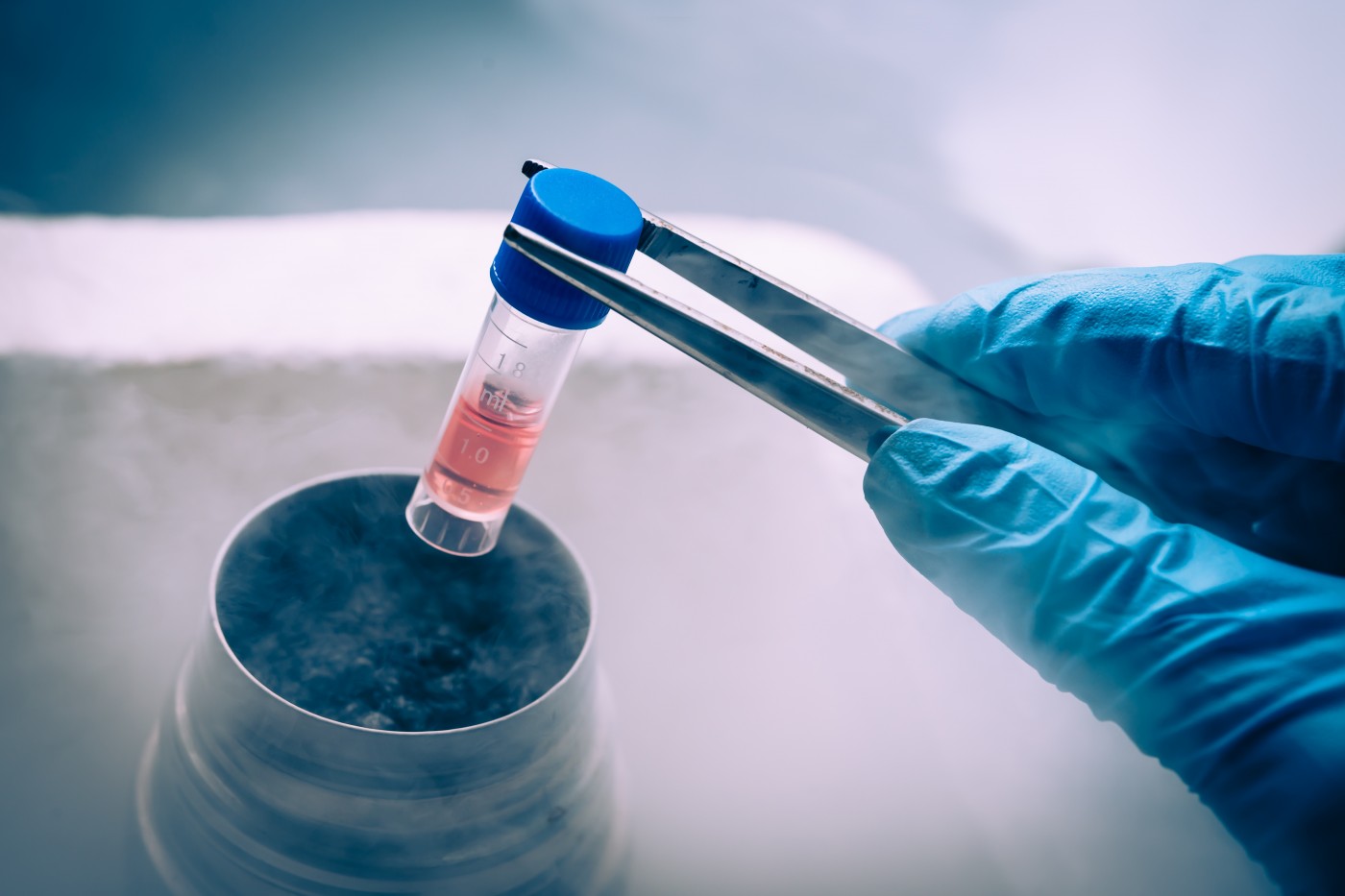
A high fraction of patients with multiple sclerosis (MS) who underwent a transplant with their own bone marrow stem cells after immunosupression therapy, show no signs of disease activity after treatment, according to a new study.
The results were presented in a talk, “Clinical experience in aggressive multiple sclerosis treatment with autologous haematopoietic stem cell transplantation,” given at the 32nd Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS), in London (Sept. 14 -17).
In MS patients, cells of the immune system, called T cells, become auto-reactive, targeting patients’ myelin, the fatty white substance that surrounds the axon of nerve cells.
In the last years, a therapeutic strategy for MS has been proposed, based on intensive immunosuppression treatment followed by autologous hematopoietic stem cell transplantation (HSCT). So, how does this work?
The idea, which already is being tested, is to first remove the harmful immune cells that attack the brain and spinal cord (immunosuppression treatment), and then use the patients’ own bone marrow stem cells to effectively re-grow their immune system. This strategy would allow the patients’ immune system to re-boot and prevent further damage in the brain and spinal cord. Several long-term follow-up observations after autologous HSCT have confirmed a great potential and efficacy for this strategy, even in aggressive forms of MS.
In his talk, Marek Smilowski, from the Department of Hematology and Bone Marrow Transplantation, Silesian Medical University, Katowice, in Poland, presented the results of a study where researchers evaluated the safety and efficacy of low-intensity immunosuppression followed by HSCT for patients with aggressive MS. In total, 89 patients with active relapsing-remitting MS (78 patients) and active secondary progressive MS (11 patients) participated in the study. The group included 49 women and 40 men, with a mean age of 38.
The study primary endpoint was to assess “No evidence of disease activity” (NEDA), defined as absence of relapses (relapse free survival – RFS), absence of confirmed disability worsening (progression free survival – PFS), and absence of radiological activity, detected by magnetic resonance (MRI-EFS).
Researchers assessed the patients regarding the Expanded Disability Status Scale (EDSS), a method to quantify disability in MS, where the higher the value, the higher the level of disability. The team showed that before autologous HSCT, the median EDSS was 5.0 and it decreased to 4.25 3 years after the treatment. The 3-year NEDA was observed in 74% of the patients.
In conclusion, the results suggest that autologous HSCT should be considered as an option for highly active MS treatment. Researchers noted, however, that the treatment must be performed according to very restrictive qualification criteria and in highly specialized hematological centers.

Eddie Nash
It is wonderful to FINALLY start seeing the OFFICIALDOM of the medical community waking up to the reality of just how well AHSCT works. Now if the entities that profit from "TREATING" MS patients will get out of the way and allow it to be an accepted treatment, maybe then the people that so desperately need it to get their lives back will have the chance. #HSCT
http://immunotherapy.nm.org/
https://www.facebook.com/groups/burthsct/
I agree.
Shirani Perera
Hi
I am a patient that have PPMS and I am supposed to get the cure in Russia by Dr.Dennis Federenco with stem Cell transplant,U.S.A. doesn't have the cure,But Russia got it.
I have a 11 year old boy born in U.S.A.,
I appreciate if you could give me a donation to get this sickness cured.
With Best wishes,
Shirani Perera.
Laura Kolaczkowski
Did the researchers share the results from the other 26% of the study participants? I wonder what happened to them and why they dropped fomcthexreported numbers. And did they mention if this was a sufficiently powered study with 89 participants? It really is interesting to see the progression for most slowed and there was the median improvement from 5.0 to 4.25 on EDSS. It can't be emphasized enough that the author of the study clearly states this is only for people with highly aggressive forms of MS and to be done in well regulated sites.
Monica Brown
I can't wait finally some promise
Eddie Nash
But NO DRUG will do what HSCT does!
http://immunotherapy.nm.org/
Monica Brown
Waiting and hoping fda helps us
Cindi Wales
The FDA is run by greedy politians, drug companies, and insurance companies. Our government has money for space and to give to other countries but not for rush approvals on treatments like HSTC! My brother was diagnosed 27 years and is Secondary-progressive-MS, and wheelchair bound. He would never be able to travel out of country for treatments.
Lori Kiker
Im 51. Maybe inmy life time they will have a cure or at least make it available to all.
Monica Brown
But where can we have it done ?
http://www.hsctstopsms.com/hsct-facilities-worldwide/
Eddie Nash
Lori Kiker HSCT:
http://immunotherapy.nm.org/
https://www.facebook.com/groups/burthsct/
Eddie Nash
Chicago:
http://immunotherapy.nm.org/
https://www.facebook.com/groups/burthsct/
Mexico at two legit places:
https://www.facebook.com/groups/mexicohsct/
Russia:
https://www.facebook.com/groups/404629779644453/
Tom
Its also good to know that stem cells are not necessary for this procedure to be effective. The stem cells facilitate in restoring the immune system quicker but do nothing to stop disease progression, the chemo suppressing the immune system and 'restarting' it is the key, a new, naive immune system. With the milder treatment and not destroying the marrow the immune system will build on its own.
Eddie Nash
Without the STEM CELLS the recovery time is much longer!
Tom
Yes Eddie. Without stem cells, recovery time is about 10-14 days vs shorter time with stem cells, 5-9 days. The point is that everyone thinks the stem cells are what make this an effective therapy to stop MS, its really the chemo.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2574697/figure/F1/
Other point is that the milder chemo regimen that does not ablate the marrow is much safer.
Eddie Nash
Tom you are absolutely correct on all points!
My point being HSCT is in all ways better that "disease MODIFYING drugs!"
As there is a stop in progression with no need for ongoing drug treatments and the associated doctors fees and drug costs!
Lalli
"highly specialized treatment centers" is a load of BS. These procedures are a lot simpler than what's done for cancer patients. I just came from Mexico (as a caregiver) and they repeated it multiple times. This can (and should) be done almost anywhere.