Brain volume loss helps in judging DMT efficacy in RRMS: Review
But MS therapy trials often fail to link disability progression and atrophy rates
Written by |

The rate of brain atrophy, or volume loss, may help in determining whether disease-modifying therapies (DMTs) are slowing disability progression in people with relapsing-remitting multiple sclerosis (RRMS), a review of data from 12 published studies found.
The analysis, “Brain Atrophy as an Outcome of Disease-Modifying Therapy for Remitting-Relapsing Multiple Sclerosis,” was published in the journal Multiple Sclerosis International.
Multiple sclerosis (MS) is caused by mistaken immune system attacks that damage myelin, a fatty substance that wraps around nerve cell fibers to protect them and help the cells communicate more effectively.
12 large MS trials from 2008 to 2019 measured changes in brain atrophy
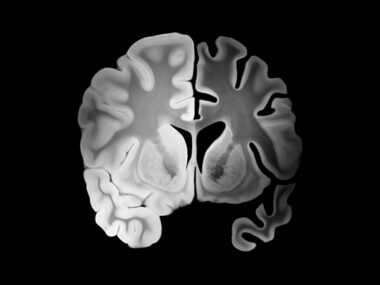
As myelin is lost and nerve cells become damaged and die, the brain shrinks, and at a faster pace in MS patients than is seen with normal aging. This atrophy, or shrinkage, is observed both in regions of damage, called lesions, and in brain regions that otherwise appear normal.
DMTs can reduce inflammatory disease activity and slow disability progression, but many clinical trials do not measure changes in brain volume or assessed how they relate to disability progression.
A team of researchers in Poland searched published studies for Phase 3 or 4 clinical trials conducted between 2008 and 2019 that reported on brain atrophy or brain volume loss as a main or secondary DMT trial goal.
From a total of 142 studies, 12 were included in the analyses, each involving more than 500 adult patients. These trials tested DMTs that either went on to be or were approved in the U.S. to treat RRMS.
“Brain atrophy, measured in clinical trials as brain volume change, is not routinely evaluated as a primary or secondary outcome,” the researchers wrote.
DMTs can reduce brain swelling and lower the number of immune cells there, causing a “pseudo” loss of brain volume lasting for up to a year. Doctors typically wait between around six months to two years on these measurements to distinguish actual shrinkage from pseudo atrophy.
Link seen between DMT use, less atrophy, slower disability progression
In all 12 studies, brain atrophy was measured at 96 weeks (nearly two years). Analysis showed that DMTs whose use resulted in a larger reduction in brain volume loss also were better at slowing disability progression in RRMS patients.
For example, Lemtrada (alemtuzumab) significantly slowed brain volume loss compared with Rebif (interferon beta-1a) in two Phase 3 studies — CARE-MS 1 (NCT00530348) and CARE-MS 2 (NCT00548405) — while reducing the risk of six-month confirmed disability by 30% to 42%.
Ocrevus (ocrelizumab) also did better than Rebif at preventing brain atrophy in OPERA I (NCT01247324) — although not in the OPERA II (NCT01412333) trial. Patients given Ocrevus in both trials had a 40% reduction in the risk of confirmed disability progression.
However, certain DMTs — for example, Mavenclad (cladribine) and Aubagio (teriflunomide) — had only a moderate effect on brain atrophy versus a placebo, although they significantly reduced the risk of disability progression.
In turn, medications such as Tecfidera (dimethyl fumarate) and Gilenya (fingolimod), both also available as generics, slowed brain volume loss in early studies, but failed to show the same effect in later studies. Later trials of these therapies also failed to show a significant slowing in disability progression.
“Our results show that [brain volume loss] in one of the components of clinical disability worsening,” the researchers concluded. Other components would be brain lesions, relapses, and disability progression, all of which contribute to a better understanding of how active and progressive a person’s disease is.
The researchers noted that one difficulty in this work is that the included studies did not always use the same MRI parameters to measure brain atrophy. To accurately compare DMTs in terms of their effect on brain atrophy, it would be important for future studies to measure brain volume loss using the same techniques and criteria, they said.
Moreover, “a longer follow-up time would likely allow brain volume changes to be a more valuable marker of neurodegeneration,” the researchers wrote.


Leave a comment
Fill in the required fields to post. Your email address will not be published.