Grey Matter Atrophy Common to All with MS and Follows Ordered Sequence, Study Finds
Written by |
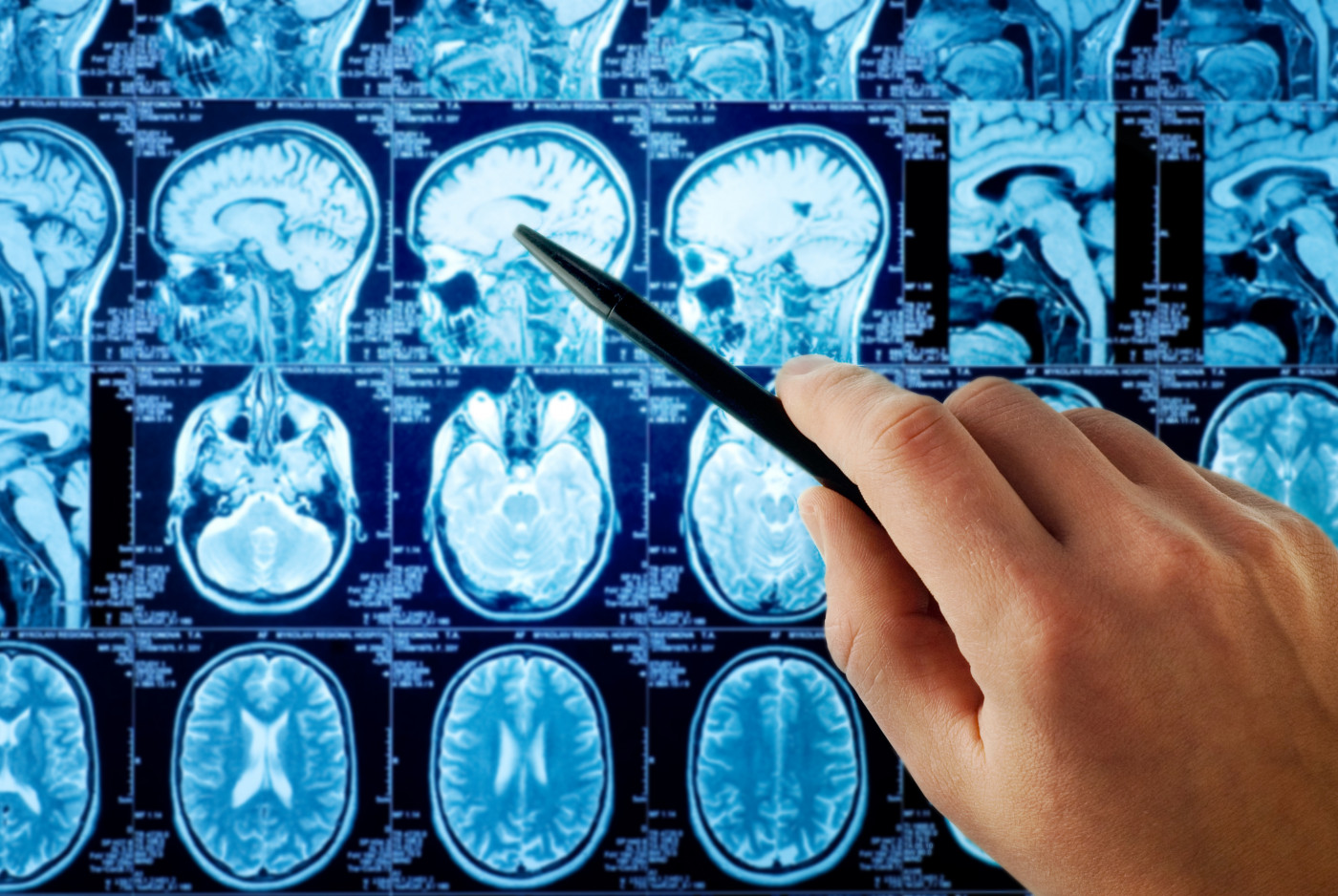
Grey matter atrophy — its loss — follows a sequential pattern that expands to involve more regions of the brain over time in all multiple sclerosis (MS) patients, regardless of their disease type, a new study reports.
Atrophy progression is also similar between relapsing-remitting multiple sclerosis (RRMS) and primary-progressive multiple sclerosis (PPMS) patients in “many key regions,” its researchers report, although three brain structures crucial to movement — the cerebellum, and the caudate and putamen (parts of the basal ganglia) — show earlier atrophy in relapsing disease, possibly as a result of greater inflammation.
The study, “Progression of regional grey matter atrophy in multiple sclerosis,” was published in the journal Brain.
MS, an inflammatory disease of the central nervous system, is characterized by brain atrophy. Studies have shown that the brains of MS patients deteriorate at a faster-than-usual rate compared to healthy people.
The brain is composed of two types of tissue — grey matter and white matter — and in MS patients, brain atrophy is mainly due to extensive grey matter atrophy. Grey matter contains most of the brain’s nerve cells, and its loss has been shown to drive MS progression and disability.
But such atrophy is not uniform across the brain, with some areas being more susceptible than others. Researchers, however, do not know if there is an order to grey matter atrophy, if it initiates and progresses in a somewhat predictable fashion from one area to another.
Researchers can trace a sequence of events through a statistical method called an event-based model, which identifies the sequence through the use of a biomarker. When the biomarker becomes abnormal, a change in the event is indicated.
This study’s team hypothesized that grey matter regions become atrophic in a consistent sequence and that this sequence will differ among different types of MS – its relapsing and progressive forms. Team members also thought it likely that increased atrophy would associate with longer disease duration and worse disability.
Researchers used a novel version of the event-based model to determine brain atrophy progression, observing the sequence of ‘events’ at which grey matter regions become atrophic in MS patients across disease types and subtypes.
Join the MS News Today forums to discuss life with MS.
They performed a retrospective analysis of 1,424 patients — 253 people with the clinically isolated syndrome, 708 with RRMS, 128 with secondary-progressive multiple sclerosis (SPMS), and 125 with PPMS — and compared the results with 203 healthy controls. Patients were recruited from seven European centers, and followed for 20 years (1996-2016); all had been part of the Magnetic Resonance in Multiple Sclerosis (MAGNIMS) project.
Magnetic resonance imaging (MRI) scans were used to measure the volume of the different brain regions, and disability levels were evaluated using the Expanded Disability Status Scale.
Applying their event-based model, researchers discovered that the first region to become atrophic in clinically isolated syndrome and RRMS patients were brain regions known as the posterior cingulate cortex (part of the brain’s limbic lobe) and precuneus (part of the parietal lobe). Atrophy was then detected in the middle cingulate cortex (also part of limbic lobe), the brainstem and thalamus (which relays sensory impulses to the cerebral cortex).
A similar, but slightly different sequence of atrophy, was seen in PPMS patients; in this group, atrophy initially involved the thalamus, cuneus (part of the occipital lobe), precuneus, and pallidum (part of the basal ganglia), followed by the brainstem and posterior cingulate cortex.
The major differences in atrophy between patients groups included cerebellum, caudate and putamen, which showed early atrophy in relapsing MS but late atrophy in PPMS patients. [The cerebellum is part of the brain’s sensory systems and essential to voluntary movement; the caudate and putamen together form the brain’s dorsal striatum.]
The authors suggest that this observation was “perhaps due to a more inflammatory milieu” in patients with relapse-onset MS.
Researchers also found that SPMS patients had the highest number of atrophic brain regions at the beginning of the study, suggesting that “secondary-progressive multiple sclerosis has more advanced neurodegeneration across multiple sclerosis phenotypes.”
But all MS patients — and distinct from people with clinically isolated syndrome — demonstrated a faster rate of grey matter atrophy than healthy controls.
Sequential grey matter atrophy was found to associate both with how long a patient had MS (disease duration), as well as with disability accumulation in RRMS patients (using EDSS measures).
Results also showed that disease-modifying therapies — which work to alleviate symptoms — and co-morbidities (other conditions) had no effect on the event-based model stages. Researchers, however, added such connections were not within the study’s scope, but were made over time. Most of these patients were “receiving the injectable first-line therapies (interferon beta and glatiramer acetate), whose effects on atrophy rates are weak,” they added.
“The data-driven staging of atrophy progression in a large multiple sclerosis sample demonstrates that grey matter atrophy spreads to involve more regions over time,” the researchers wrote.
“The event-based model has a potential for clinical use,” they added, suggesting that the “sequence of atrophy progression can be used to score patients during multiple sclerosis automatically.”


Heidi
Does Lemtrada reduce brain atrophy I wonder? I notice most of the study subjects had used only used basic (first line) DMDs.
Karen Smith
What kind of disability correlates to this brain atrophy? I’m not knowledgeable about what the areas of the brain mentioned in this study actually control.
Donna Bretthauer
This is the best article. It is easy to understand.